Left ventricular reverse remodeling (LVRR), defined as reduction of end-diastolic and end-systolic dimensions and improvement of ejection fraction, is associated with the prognostic implications of cardiac resynchronization therapy (CRT). The time course of LVRR remains poorly characterized. Nevertheless, it has been suggested that it occurs ≤6 months after CRT.
ObjectiveTo characterize the long-term echocardiographic and clinical evolution of patients with LVRR occurring >6 months after CRT and to identify predictors of a delayed LVRR response.
MethodsA total of 127 consecutive patients after successful CRT implantation were divided into three groups according to LVRR response: Group A, 19 patients (15%) with LVRR after >6 months (late LVRR); Group B, 58 patients (46%) with LVRR before 6 months (early LVRR); and Group C, 50 patients (39%) without LVRR during follow-up (no LVRR).
ResultsThe late LVRR group was older, more often had ischemic etiology and fewer patients were in NYHA class ≤II. Overall, group A presented LVRR between group B and C. This was also the case with the percentage of clinical response (68.4% vs. 94.8% vs. 38.3%, respectively, p<0.001), and hospital readmissions due to decompensated heart failure (31.6% vs. 12.1% vs. 57.1%, respectively, p<0.001). Ischemic etiology (OR 0.044; p=0.013) and NYHA functional class
ConclusionsLate LVRR has better clinical and echocardiographic outcomes than no LVRR, although with a suboptimal response compared to the early LVRR population. Ischemic etiology and NYHA functional class
A remodelagem inversa do ventrículo esquerdo (RIVE), definida pela redução das dimensões telediastólicas e telessistólicas e pela melhoria da fração de ejeção, tem sido associada às implicações prognósticas da terapia de ressincronização cardíaca (TRC). A evolução temporal da RIVE permanece um processo pouco caracterizado. No entanto, tem sido sugerido que ocorrerá num período ≤6 meses após TRC.
ObjetivosCaracterizar a longo prazo a evolução ecocardiográfica e clínica dos pacientes (P) com evidência de RIVE, num período >6 meses após TRC, e identificar preditores de uma resposta de RIVE tardia.
MétodosCento e vinte e sete P consecutivos, após implantação bem-sucedida de TRC, foram divididos em três grupos, de acordo com a resposta de RIVE: grupo A, 19P (15%) com RIVE após seis meses (RIVE tardia); grupo B, 58P (46%) com RIVE antes dos seis meses (RIVE precoce) e grupo C, 50P (39%) sem RIVE durante o follow-up (sem RIVE).
ResultadosO grupo da RIVE tardia era mais velho, tinha mais etiologia isquémica e menos P em classe NYHA≥III. Globalmente, o grupo A apresentou um grau de RIVE entre os grupos B e C. O mesmo ocorreu em relação ao grau da resposta clínica (68,4 versus 94,8 versus 38,3%, respetivamente, p<0,001) e às readmissões hospitalares por descompensação da insuficiência cardíaca (31,6 versus 12,1 versus 57,1%, respetivamente, p<0,001). A etiologia isquémica (OR 0,044; p=0,013) e a classe funcional
ConclusõesA RIVE tardia tem uma melhor resposta clínica e ecocardiográfica do que a ausência de RIVE, embora com uma resposta subótima, quando comparada com a população com RIVE precoce. A etiologia isquémica e uma classe NYHA
Cardiac resynchronization therapy (CRT) has become an effective non-pharmacological treatment for patients with impaired left ventricular ejection fraction (LVEF), prolonged QRS duration and New York Heart Association (NYHA) functional class ≥III symptoms of heart failure (HF) despite optimal medical therapy. Large multicenter clinical trials have shown that CRT can mitigate HF symptoms, improve exercise capacity and quality of life, reduce the incidence of hospitalizations due to HF and decrease mortality.1–4 Most of these benefits are thought to be related to left ventricular (LV) reverse remodeling (LVRR), a response characterized by a reduction in LV end-diastolic (LVEDD) and end-systolic (LVESD) dimensions and volumes and improvement of LV ejection fraction (LVEF).5–7 The time course of this phenomenon remains poorly characterized. It has been suggested that the structural and functional changes associated with LVRR occur early after CRT and are more pronounced before the six-month landmark, a time by which the extent of reverse remodeling has been shown to predict long-term prognosis in these patients.6–9 Nevertheless, a variable proportion of eligible patients fail to respond initially but appear to develop LVRR after six months following CRT implantation. This population with late LVRR, who are not included in the already known proportion (around 30%) of non-responders to CRT, correspond to a different spectrum of individuals that, to the best of our knowledge, has never been directly analyzed in a prospective study. To overcome this limitation, the present study characterizes the timing of LVRR occurrence and assesses the long-term echocardiographic and clinical evolution of patients with LVRR occurring >6 months after CRT implantation. We also sought to identify potentially related clinical characteristics that might predict a late response to CRT.
MethodsThis is a single-center analysis of patients who underwent successful CRT device implantation. A total of 127 consecutive CRT recipients were included and patient data were prospectively collected in the information system of our cardiology department and analyzed. Patients were selected for CRT if they met currently recommended criteria: (1) LVEF <35%; (2) symptoms of HF, defined as NYHA class II–IV despite optimal medical therapy; and (3) QRS duration ≥120 ms. Patients were classified as ischemic in the presence of significant coronary artery disease (>50% stenosis of two or more epicardial vessels or >50% left main or proximal left anterior descending coronary artery stenosis on coronary angiography and/or a history of previous myocardial infarction or revascularization). Other patients were classified as non-ischemic. All leads were placed transvenously via the subclavian and cephalic route using fluoroscopy. The right ventricular lead was positioned in the apex or mid septum. The LV lead was placed with an over-the-wire system in the posterolateral or lateral tributary vein of the coronary sinus depending on the ability to cannulate the veins, pacing threshold, or diaphragmatic stimulation. The standard settings included an atrioventricular (AV) delay of 100 ms (sensed) and 130 ms (paced), with DDD or DDDR mode and standard lower (50 beats/min) and upper (120–130 beats/min) pacing rates. Extensive demographic and clinical data, including death, NYHA class and hospitalization for worsening HF or CRT dysfunction, were collected from medical records. Transthoracic two-dimensional echocardiographic information was assessed at baseline, six months after CRT device implantation and thereafter once a year until the end of follow-up. In this study, LVRR was defined as an increase >10% in LVEF over baseline, combined with a >10% reduction in LVEDD and LVESD. Echocardiographic cardiac structure and function were assessed using a commercially available ultrasound system (Vivid 7 and Vivid E9; GE Vingmed Ultrasound, Horten, Norway) equipped with a 3.5-MHz transducer. LVESD, LVEDD and LVEF were determined according to standard techniques and digitally stored for offline analysis in cine-loop format. Interventricular mechanical delay was calculated by pulmonary and aortic pulsed Doppler measurement of the difference in time from QRS onset to onset of flow in the pulmonary and aortic outflow tracts, respectively. LV intraventricular dyssynchrony was assessed offline using a previously validated software package (EchoPAC PC-2D strain; GE Healthcare, USA). The cohort was divided according to the time course of LVRR response into three groups: Group A, presence of LVRR after >6 months (late LVRR); Group B, with LVRR before 6 months (early LVRR); and Group C, without LVRR during follow-up (no LVRR). Follow-up data were obtained by review of medical records, outpatient clinical visits, and telephone contact. Ethical and hospital permission were obtained from the appropriate local authorities.
Statistical analysisSPSS version 21 software (SPSS Inc., Chicago, Illinois) was used for computation. Data are expressed as means ± standard deviation for continuous variables and as frequencies and percentages for categorical variables. Data distribution was tested for normality using the Kolmogorov–Smirnov or Shapiro–Wilk test as appropriate. Missing patient-level covariates were assumed to be missing and no imputation was performed. Baseline characteristics and outcomes were compared using the chi-square test or Fisher's exact test, when appropriate, for categorical variables and the Student's t test or the Mann–Whitney test for continuous variables. Univariate and multivariate logistic regression analysis was used to examine for potential predictors of a delayed LVRR response. A value of p<0.05 was considered statistically significant.
ResultsPatient baseline characteristicsOverall, the study population consisted of 127 consecutive patients who underwent successful CRT implantation (aged 63.9±10.7 years, 74.8% male). The mean follow-up was 48±36 months (median 37 months). Baseline patient characteristics are shown in Table 1. The majority of the cohort (60.6%) had non-ischemic cardiomyopathy.
Baseline clinical and echocardiographic characteristics.
| Baseline | All patients n=127 | Late LVRR n=19 | Early LVRR n=58 | No LVRR n=50 | p |
|---|---|---|---|---|---|
| Age (years) | 63.9 | 70±10.7 | 62.2±11.0 | 63.3±8.9 | 0.026 |
| Male (%) | 74.8 | 73.7% | 69.0% | 82.0% | NS |
| Etiology (%) | |||||
| Ischemic | 29.9 | 42.1% | 13.8% | 44% | <0.001 |
| Non-ischemic | 60.6 | 31.6% | 77.6% | 52% | <0.001 |
| Mixed | 9.5 | 21.1% | 3.4% | 4.0% | 0.041 |
| Baseline ECG | |||||
| LBBB | 79.5 | 76.5% | 92.6% | 76% | 0.038 |
| QRS (ms) | 170.02 | 164±26.78 | 176.29±29.75 | 164.85±31.83 | NS |
| Sinus rhythm | 66.1 | 57.9% | 74.5% | 59.6% | NS |
| NYHA class ≥III | 92.1 | 78.9% | 96.6% | 92% | 0.021 |
| LVESD (mm) | 61.3 | 70±10.7 | 62±11.01 | 63±8.9 | NS |
| LVEDD (mm) | 74.5 | 74±9.4 | 72±22 | 77±9.0 | 0.048 |
| LAD (mm) | 47.9 | 50±10.6 | 46±9.10 | 50±12.9 | NS |
| LVEF | 24.8% | 26.3% | 23.7% | 25.8% | NS |
| Pitzalis (ms) | 210.8 | 202±115.5 | 240±129.5 | 168±128.6 | 0.049 |
| IVMD | 44.5 | 47±28.2 | 51±27.6 | 37±30.5 | NS |
| LVlat-IVS delay (ms) | 134.9 | 130±113.1 | 119±92.7 | 135±95.3 | NS |
IVMD: interventricular mechanical delay; LAD: left atrial diameter; LBBB: left bundle branch block; LVEDD: left ventricular end-diastolic diameter; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic diameter; LVlat-IVS: left ventricular lateral wall to interventricular septum; LVRR: left ventricular reverse remodeling; NYHA: New York Heart Association; Pitzalis: LV dyssynchrony by the Pitzalis method. A mixed etiology was defined as the presence of both ischemic and non-ischemic causes.
Regarding differences between groups, late LVRR patients were older than those in Groups B and C (70 years vs. 62.2 years vs. 63.3 years, respectively, p=0.026) and fewer were in NYHA class ≥III (78.9% vs. 96.6% vs. 92%, respectively, p<0.021). Group B, with an early favorable CRT response, were younger than those in Groups A and C (62.24 years vs. 70 years vs. 63 years, respectively, p=0.026), less often had ischemic etiology (13.8% vs. 42.1% vs. 44%, respectively, p<0.001), included more patients with left bundle branch block (LBBB) (92.6 vs. 76.5 vs. 76, respectively, p=0.038) and showed more LV dyssynchrony (240±129.5 vs. 202±115.5 vs. 168±128.6, respectively, p=0.049) as evaluated by the Pitzalis method. Group C, the non-responders, had more patients with ischemic etiology than Groups A and B (44 vs. 13.8 vs. 42.1, respectively, p<0.001), and had greater LVEDD (77±9.02 vs. 74±9.38 vs. 72±22 mm, respectively, p=0.048) and less previous LV dyssynchrony (168±128.6 vs. 202±115.5 vs. 240±129.5 ms, respectively, p=0.049), as assessed by the Pitzalis method. No statistically significant differences were detected in other electrocardiographic or echocardiographic parameters.
Echocardiographic and clinical changes after cardiac resynchronization therapyComparison between groups of echocardiographic data from the last evaluation during follow-up is displayed in Table 2. After CRT implantation, LVRR occurred in 77 (60.6%) patients. The time course of LVRR was different within this population, with 19 patients having late LVRR (Group A), and 58 patients having evidence of early LVRR (Group B). A significant difference was noticed between groups in terms of the extent of LVRR response. At the last echocardiographic evaluation, compared to Groups A and C, Group B presented lower LVEDD (66±12.33 vs. 68±10.05 vs. 77±11.12 mm, respectively, p<0.001), lower LVESD (49±12.9 vs. 53±11.8 vs. 63±11.0 mm, respectively, p<0.001), higher LVEF (43±9.4 vs. 41±7.73 vs. 27±6.3%, respectively, p=<0.001) and smaller left atrial diameter (44±9.3 vs. 54±10.0 vs. 50±11.2, respectively, p<0.001).
Echocardiographic characteristics at follow-up.
| Follow-up | All patients n=127 | Late LVRR n=19 | Early LVRR n=58 | No LVRR n=50 | p |
|---|---|---|---|---|---|
| LVESD (mm) | 53.53±13.56 | 53±11.80 | 49±12.93 | 63±11.04 | <0.001 |
| LVEDD (mm) | 69.31±12.45 | 68±10.05 | 66±12.33 | 77±11.12 | <0.001 |
| LAD (mm) | 47.38±10.76 | 54±10.04 | 44±9.26 | 50±11.22 | <0.001 |
| LVEF (%) | 38.48±11.04 | 41±7.73 | 43±9.35 | 27±6.32 | <0.001 |
LAD: left atrial diameter; LVEDD: left ventricular end-diastolic diameter; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic diameter; LVRR: left ventricular reverse remodeling.
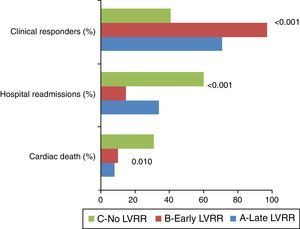
Regarding clinical outcomes, during the mean follow-up of 48±36 months, both groups A and B presented better clinical status than Group C (Figure 1). The clinical response to CRT, defined as a sustained improvement in NYHA functional class, was more evident in group B than in groups A and C (94.8% vs. 68.4% vs. 38.3%, respectively, p<0.001). Group B subjects were also less often hospitalized for worsening of HF symptoms (12.1% vs. 31.6% vs. 57.1%, respectively, p<0.001). Group C showed higher cardiac mortality compared to Groups A and B (28.6% vs. 5.6% vs. 7.3%, respectively, p=0.010).
Predictors of late left ventricular reverse remodelingIn order to identify characteristics that might have an impact on the time course of LVRR after CRT, a logistic regression analysis was performed to predict a late LVRR response (Table 3). Ischemic etiology (odds ratio [OR] 0.044; p=0.013) and NYHA functional class ≤II (OR 0.056; p=0.063) were the baseline variables with the highest predictive value for a late LVRR response to CRT.
DiscussionThe major findings of the present study, which focused on the impact of the time course of LVRR after CRT, are as follows: first, the majority of patients (60.6%) presented evidence of LVRR during long-term follow-up; second, 14.9% showed LVRR only six months after CRT; third, late LVRR patients were older and more often had ischemic etiology, and fewer were in NYHA class ≥III; fourth, late LVRR was significantly associated with better clinical and echocardiographic outcomes than no LVRR, although with a suboptimal response compared with the early LVRR population; and finally, ischemic etiology and NYHA functional class ≤II were independent predictors of late LVRR.
Several studies have addressed the favorable impact of CRT in mid- and long-term follow-up, which usually occurs in the first six months after implantation.10–15 The underlying mechanism responsible for this early improvement seems to be the LVRR response initiated soon after biventricular pacing.5–7 Van de Veire et al., analyzing 49 patients scheduled for CRT, documented a decrease of ≥15% in LV end-diastolic volume in 47% of patients in the first 48 hours after device implantation.15 Nina et al., in a review of 222 consecutive patients with HF implanted with a CRT system, reported a significant increase in LVEF and a decrease in LV end-diastolic volume (LVESV) acutely after implantation followed by an additional improvement in these parameters at six-month follow-up.16 Wang et al., in a study of 30 patients scheduled for CRT, revealed a decrease of ≥15% in LVESV before six months in 60% of the population.17 Also, Sanderson et al., in a report of 30 patients scheduled for CRT, revealed an increase of ≥15% in LVESV at three months after implantation in 56.7% of the population.18 However, to the best of our knowledge, no reports have focused on LVRR documented only after six months. There are no studies demonstrating differences in the long-term prognosis of this group compared with early reverse remodeling. Pires et al., in a related study of 313 HF patients from the MIRACLE-ICD trial, compared early (1–3 months) and late (six months) clinical response after CRT and correlated it with the presence or absence of LVRR, but no significant difference was found.19
Despite being a recognized therapy with a favorable influence on NYHA functional class, quality-of-life scores, rehospitalizations and cardiac death, there are a significant proportion of patients, estimated at 30%, who are considered clinical non-responders to CRT.20–22 This population has been extensively studied in previous reports and many factors have been proposed as potential contributors to a lack of response. In this study we found that a delayed reverse remodeling response after six months shares some of the variables that are present in non-responders.
Patients with an LVRR response, in fact, also had less conventional electrical dyssynchrony (i.e. shorter QRS interval), even though not statistically significant, and less severe mechanical dyssynchrony evaluated by echocardiography at baseline. Although the PROSPECT trial showed modest sensitivity and specificity of echocardiographic parameters of dyssynchrony,23 subanalysis including 286 patients from the original PROSPECT cohort that focused on three simple preprocedural measures of dyssynchrony reported a strong association of their parameters with more extensive reverse remodeling at 6-month follow-up.24 LBBB is clearly more evident in early LVRR patients.25,26 In our study, Groups B and C had a higher incidence of ischemic etiology. Several previous studies have shown less clinical and LV function improvement in ischemic patients, probably due to the presence of myocardial scar tissue in the vicinity of the pacing lead, limiting the extent of LVRR regardless of the extent of baseline dyssynchrony.20–22 Pressure and volume overload associated with LV involvement typical of HF patients leads to gradual fibrosis, left atrial (LA) enlargement and subsequently mitral regurgitation. In this report, as stated in other studies, an increased in LA size was also correlated with late or no remodeling.27,28
Those who responded early with reverse remodeling were more symptomatic and also had higher LV systolic and diastolic dimensions at baseline, suggesting that sicker patients have more to benefit from CRT. There are other factors that could help to explain the difference in LVRR. Identification and treatment of reversible causes in the first six months after CRT, such as AV and VV delays and suboptimal HF therapy, could transform a late into an early response. Furthermore, factors such as the position of the LV lead, optimization of device programming and the presence of rhythm abnormalities could be related to a delayed response, but were not analyzed in this report.
Regarding clinical outcomes during follow-up, the achievement of LVRR, regardless of duration of CRT, was followed by symptomatic and prognostic improvement as demonstrated by a sustained decrease in NYHA functional class, fewer hospitalizations due to HF worsening and less cardiac deaths compared to non-responders. Potential underlying factors could be the restoration of inter- and intraventricular synchrony and improvements in contractility and hemodynamics. These results are in line with previously described long-term positive outcomes of reverse remodeling obtained by CRT.5–7 However, analyzing early and late LVRR responses, it is clear that clinical benefits are more pronounced in the early group. This could be explained, in part, by the less intense LVRR response achieved in Group A, with a smaller impact on LV dimensions and LVEF compared with Group B. Nevertheless, long-term outcomes of late LVRR patients are significantly better than non-responders.
The mechanism of a delayed response to CRT is complex and probably multifactorial, with no conclusive explanation currently available. Proper selection of a suitable subset of patients is mandatory, since it appears that sicker HF patients may respond better to CRT. According to our findings, ischemic etiology and NYHA functional class
LimitationsThe present study had some limitations. First, it was a single-center retrospective cohort, albeit representative of real-world clinical practice. Second, the definition of LVRR, also used in previous studies,30 required a cut-off of 10% reduction in LV dimensions and an increase of 10% in LVEF. Other studies were stricter and used a 15% threshold.29,30 Third, clinical response was based only on improvement in NYHA class and did not include assessment of functional capacity or quality of life scores. Fourth, echocardiographic parameters depend on the operator and there will always be a degree of intra- and interobserver variability. Fifth, other parameters such as position of LV lead, device programming, presence of rhythm abnormalities and levels of brain natriuretic peptide (BNP) were incomplete and thus were not considered for the present study. Finally, because of the modest number of patients studied, the current findings regarding late LVRR predictors need confirmation in large-scale prospective studies.
ConclusionsOur data confirm, like other reports, that CRT is associated with favorable clinical and echocardiographic responses during long-term follow-up. This study is the first of this kind to analyze the time course of LVRR and to characterize the impact of a delayed reverse remodeling response in long-term follow-up. Late LVRR is significantly associated with better clinical and echocardiographic outcomes than no LVRR, although with a suboptimal response compared to the early LVRR population. Improvement in HF symptoms and survival after CRT are proportional to the timing and extent of LVRR response. Among all baseline parameters studied, ischemic etiology and NYHA functional class ≤II were identified as predictors of late LVRR. Better selection of suitable patients may potentially improve the rate of early CRT responders.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of interestThe authors have no conflicts of interest to declare.