By May 2020, SARS-CoV-2 had caused more than 400 000 deaths worldwide. Initially, hydroxychloroquine was a treatment option for COVID-19. More recent studies have questioned its safety and efficacy and, until stronger evidence is available, it was suspended from therapy protocols. We describe our experience treating COVID-19 Portuguese pediatric patients with hydroxychloroquine, having applied a protocol for monitoring cardiac toxicity.
MethodsAn observational retrospective study of COVID-19 pediatric patients, admitted from March to April 2020 and treated with hydroxychloroquine. Cardiotoxicity was assessed using ECG recordings and corrected QT-time (QTc). Patients were classified into risk-groups depending on QTc value: normal, slightly elevated or severely elevated (>500 ms).
ResultsTotal of 14 patients, with a median age of 10 years [four months; 17 years], treated with hydroxychloroquine for a median of five days. Hydroxychloroquine was used in monotherapy in six patients (mainly mild disease with comorbidities), and in association with lopinavir/ritonavir (3) and azithromycin (5) in moderate to severe disease. Other QT-prolonging therapies were used in five patients: oseltamivir (3), omeprazole (1), morphine (1) and ketamine (1). At 48 hours of treatment, two patients temporarily suspended hydroxychloroquine due to QTc prolongation (>500 ms). All patients completed the whole treatment. No other side effects or deaths occurred.
ConclusionClinical trials are evolving to define hydroxychloroquine effectivity and safety. Our considerable pediatric population supports the need for cardiotoxicity monitoring during therapy but suggest its use seems to be safe in COVID-19 pediatric patients, even in association with other QT-prolonging therapies.
A hidroxicloroquina foi inicialmente uma das opções terapêuticas na Covid-19. Descreve-se o tratamento com hidroxicloroquina em doentes Covid-19 pediátricos, tendo aplicado um protocolo de monitoração cardíaca pelo seu potencial arritmogénico.
MétodosEstudo observacional retrospetivo de doentes pediátricos com Covid-19, internados de março a abril 2020, medicados com hidroxicloroquina. A monitoração cardíaca foi realizada por eletrocardiogramas regulares e cálculo do intervalo QT corrigido durante o tratamento. Os doentes foram classificados consoante o valor de QTc: normal, moderadamente aumentado ou muito aumentado (>500 msg).
ResultadosTotal de 14 doentes, com mediana de 10 anos [4 meses; 17 anos], medicados com HCQ durante uma mediana de 5 dias em doentes com pneumonia ou comorbilidades. A monoterapia foi realizada em 6 doentes, 4 com fatores de risco, e em associação com lopinavir/ritonavir (3) e azitromicina (5) na doença grave e moderada. Foram ainda usados fármacos capazes de prolongar o intervalo QT: oseltamivir (3), omeprazol (1), cetamina e morfina (1) em 5 doentes. Após 48 horas de terapêutica, dois doentes apresentaram intervalo QTc muito aumentado, condicionando suspensão temporária do fármaco. Todos os doentes concluíram o tratamento sem outros efeitos adversos.
ConclusãoA HCQ permanece em ensaios clínicos para avaliação da sua efetividade e segurança. A nossa amostra considerável em doentes pediátricos apoia a necessidade de monitoração de toxicidade cardíaca, mas sugere na população estudada, mesmo na associação com outros fármacos que prolongam o intervalo QT, a segurança de sua utilização.
In December 2019, a novel betacoronavirus was found to be responsible for a cluster of atypical pneumonia cases in the Hubei Province in China.1 A few months later, the new betacoronavirus, subsequently named SARS-CoV-2, causing the COVID-19 disease (coronavirus disease 2019), had spread to various other countries throughout the world.2 At the end of May 2020, more than 400000 deaths had been reported.3 Although the pediatric population seemed to have been less affected by the infection than adults and the clinical presentation as well as evolution were generally less severe, critical cases nonetheless, occurred.4 Since the beginning of the pandemic, an association between COVID-19 and adverse cardiovascular outcomes had been described.5 Just like other coronavirus, SARS-CoV-2 seems to use angiotensin converting enzyme II (ACE2) as an entry receptor in human cells.6 ACE2 expression in vascular tissue plays a major role in heart function,7 and is a negative regulator of Ang II-induced myocardial hypertrophy, fibrosis, and diastolic dysfunction.8
The cardiac pathophysiology of COVID-19 is not fully understood. It has been hypothesized that one of the contributions to COVID-19-associated myocardial damage and inflammation is the interaction between SARS-CoV-2 and ACE2, expressed in respiratory tract, heart and endothelial cells.9 The direct tissue infection can lead to cellular inflammation. A systemic inflammation is also induced by SARS-CoV-2, with a consequent cytokine storm (with elevation of IL-6) and increased oxidative stress. This pro-inflammatory state may be associated with higher risk of cardiac arrhythmias and myocardial injury.10–12
Currently, there is no antiviral treatment with proven efficacy, but several ongoing trials are assessing the efficacy of antiviral agents such as hydroxychloroquine, lopinavir-ritonavir, favipravir and remdesivir and of immunomodulators, including tocilizumab and anakinra. Data on children and adolescent affected by COVID-19 remain scarce. The management of COVID-19 patients is based on limited data and evolves rapidly as clinical knowledge emerges. Although interim guidance has been issued by the World Health Organization (WHO),12 a number of different treatment guidelines have been published by many scientific societies around the world since the beginning of the pandemic.13–15
Among the proposed drugs for COVID-19 treatment, hydroxychloroquine (HCQ) has arisen as one of the most used drugs for the treatment of moderate to severe COVID-19 patients.16
The cardiac impact of the use of HCQ as off label therapy in COVID-19 is yet unknown17 as the optimal protocol for monitoring cardiac toxicity has not yet been determined.18 Although the pediatric population is less prone to developing myocardial infarction and heart failure, the risk of having an undiagnosed congenital rhythm disorder is greater than in the adult population.
Our center is a national reference hospital for COVID-19 pediatric patients and has been responsible for the management of a major portion of pediatric severe cases in Portugal. Since we admitted the first pediatric patient on 7 March 2020, HCQ has been used in the treatment of patients, among other experimental drugs (e.g. lopinavir/ritonavir, remdesivir), with the caveat explained to parents that its efficacy had not yet been proven but that HCQ had been included in several treatment protocols with pediatric COVID-19 patients throughout the world. Since the beginning, we formed a close relationship with the Pediatric Cardiology Department to determine the safest way to utilize HCQ. A protocol was adapted from a similar already published protocol on Mayo Clinic Proceedings18 and applied in all patients admitted to our unit and in whom HCQ treatment was proposed.
We aimed to describe the cardiac outcome in a population of pediatric patients diagnosed with COVID-19 and medicated with hydroxychloroquine since the implementation of a protocol for monitoring cardiac and rhythm toxicity in our center. As far as we know, this is the first report of HCQ used in pediatric patients, and the first to be conducted in Portugal.
MethodsWe included all pediatric patients (<18 years) admitted to a tertiary pediatric hospital with the diagnosis of COVID-19 and medicated with HCQ, between March and April 2020 (two months). Data was collected from electronic medical records and photographs electrocardiogram (ECG) records.
Collected demographic and clinical data included age, gender, clinical severity, comorbidities and other concomitant therapies. All ECGs were assessed. Therapy dosage, duration and need for suspension were also analyzed.
For a positive diagnosis of COVID-19, the following were considered: the detection of SARS-CoV-2 by reverse-transcriptase polymerase chain reaction in respiratory swabs (oro and nasopharynx) and/or respiratory tract secretions samples and suggestive signs of the infection in thoracic computerized tomography (CT).
Disease severity and need for treatment with HCQ were assessed according to the local hospital guidelines. Patients were considered as having mild disease if they presented with fever, cough, anorexia, myalgias, sore throat, nasal obstruction or headache and there were no typical findings on the chest CT; moderate if presented with pneumoniae without hypoxemia (peripheral oxygen saturation >93%) or other signs of severity; severe if presented with pneumoniae and signs of respiratory distress (central cyanosis or tachypnea: >70 bpm in <12 months and >50 bpm in those >12 months; severe intercostal retraction, expiratory groan or nasal flaring) or other signs of severity (no oral intake, alteration in mental status or seizures) and critical (fulfilling criteria for acute respiratory distress syndrome, sepsis or septic shock).
In all patients with pneumoniae, other viral respiratory pathogens and atypical pneumoniae agents were searched by polymerase chain reaction on nasal/respiratory secretions.
Risk factors for potential severe disease included immunosuppressed patients (primary immunodeficiency, HIV infection with detectable viral load, solid organ or hematopoietic transplant recipient and treatment with immunosuppressive drugs), congenital cardiopathy, chronic pulmonary disease, dialysis patients, sickle cell anemia, type I diabetes with bad metabolic control, neuromuscular disease, severe encephalopathy patients, hereditary metabolic disorder, short bowel syndrome and severe malnutrition.
Hydroxychloroquine was indicated in the treatment of moderate, severe and critical disease and also in mild disease with known risk factors. The HCQ dose was 6.5 mg/kg/dose BID on the first day, and 3.25 mg/kg/dose, BID on subsequent days. Treatment duration was defined based on clinical evolution, between five and 10 days. Written informed consent was obtained from the parents of all treated patients.
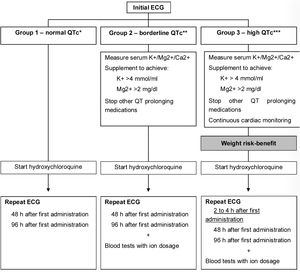
A protocol for cardiac monitoring in COVID-19 patients treated with HCQ alone or in combination with other therapies was adapted from a Mayo Clinic Proceedings’ protocol and applied (Figure 1).18 Monitoring was performed using electrocardiogram (ECG), reviewed by a pediatric cardiologist and serum electrolytes levels. A pre-treatment ECG was performed in all diagnosed COVID-19 patients. The corrected QT interval (QTc) was calculated manually, using DII and V5 derivations and the Bazett or Fridericia formulas. QTc was considered normal if it was <460 ms in pre-puberty children, lower than 470 ms in male adolescents, and lower than 480 ms in female adolescents.
Diagram of pre-treatment cardiac assessment and cardiac toxicity monitorization during treatment.18 Legend: * Normal QTc – A normal value was considered according to age and gender. Before puberty QTc<460 ms; after puberty, in male QTc<470 ms and female QTc<480 ms. ** Borderline QTc – values above normal but lower than 500 ms. *** Hight QTc – values above 500 ms. Adapted from Giudicessi et al.18 Guidance on Mayo Clinic Proceedings doi:10.1016/j.mayocp.2020.03.024.
According to the QTc, patients were classified in one of three groups: Group 1 for patients with normal QTc; Group 2 for patients with increased QTc but <500 ms and Group 3 for patients with QTc >500 ms, regardless of age. Patients treated with HCQ underwent therapy toxicity monitoring at 2-4 h, 48 h or 96 h of therapy depending on the classification group (Figure 1). For patients in Group 2 and Group 3, other factors known to prolong QT interval were investigated. Blood electrolytes were measured, and potassium, calcium and magnesium levels were corrected, as applicable. Other medications known to cause QT prolongation were suspended, when possible. In the case of patients in Group 3, the risk-benefit of treatment had to be assessed. An elevation in QTc interval >60 ms compared to the pretreatment value or a value >500 ms at any point resulted in re-assessment of the case and discussion regarding therapy suspension/continuation.
ResultsA total of 14 patients were admitted during the study period and treated with HCQ, alone or in combination with other medicines. The median age was 10 years, ranging from 4 months to 17 years old and the most prevalent gender was female (nine patients), as described in Table 1.
Demographic and clinical characteristics of the patients under study.
| Case nr | Clinical severity | Age (years) | Gender | Comorbidities | Chest CT findings (n=10) | Need for oxygen (n=8) | Coinfection |
|---|---|---|---|---|---|---|---|
| 1 | Mild (n=4) | 11 | Female | yes | no | no | no |
| 2 | 13 | Female | no | no | no | no | |
| 3 | 8 | Female | yes | no | no | no | |
| 4 | 16 | Female | yes | no | no | no | |
| 5 | Moderate (n=2) | 16 | Female | no | Pneumonia | no | no |
| 6 | 4 | Female | no | Pneumonia | no | Mycoplasma pneumoniae+Rhinovirus | |
| 7 | Severe (n=8) | 0 | Male | no | Pneumonia | no | no |
| 8 | 0 | Female | yes | Pneumonia | yes | no | |
| 9 | 11 | Male | no | Pneumonia | yes | Mycoplasma pneumoniae+Rhinovirus | |
| 10 | 15 | Male | yes | Pneumonia | yes | Mycoplasma pneumoniae | |
| 11 | 1 | Female | no | Pneumonia | yes | Metapneumovírus | |
| 12 | 17 | Male | no | Pneumonia | yes | Rhinovirus | |
| 13 | 9 | Male | no | Pneumonia | yes | no | |
| 14 | 6 | Female | no | Pneumonia | yes | Chlamydia pneumoniae+Rhinovirus |
The interval between symptom onset and diagnosis was two to 15 days, with a mean of 6.2 days. All patients started therapy within 24 hours of diagnosis, apart from two patients, with a maximum delay of 72 hours.
Five patients had comorbidities: juvenile idiopathic arthritis under immunosuppressive therapy – etanercept (1) and mycophenolate mofetil (1); sickle cell anemia (1) and chronic respiratory diseases – bronchopulmonary dysplasia (1) and chronic tracheostomy (1). Regarding clinical severity, most patients were considered severe (8), all with evidence of pneumonia on the chest CT, and seven needed supplemental oxygen. Two patients classified as moderate showed pneumonia on CT-scan, none with hypoxemia. Mild disease was found in four patients with upper respiratory tract symptoms and no radiological signs of pneumonia.
Coinfection was identified in six patients, in some cases with more than one microorganism. Mycoplasma pneumoniae (3), Chlamydia pneumoniae (1), adenovirus (1), metapneumovirus (1) and rhinovirus (1) were reported.
No patient was diagnosed with acute respiratory distress syndrome, needed ventilatory support or admission to a pediatric intensive care unit. No deaths were recorded. All patients were discharged home without chronic complications, except one infant with bronchopulmonary dysplasia who required a higher level of oxygen than before COVID-19.
Treatment and cardiac monitoring. Treatment length with HCQ was five days in 12 patients and seven days in the two younger patients due to their slower, although favorable, clinical evolution.
Hydroxychloroquine as monotherapy was adopted in six children: all four mild patients, one moderate adolescent and a severe one year old child, as described in Table 2. Other medicines were combined with HCQ in eight patients: lopinavir/ritonavir in three severe cases (diagnosed before disease day 4), and azithromycin in four severe and one moderate cases. Nine patients also received other antibiotics as ceftriaxone (6), cefotaxime, clindamycin, amikacin, ceftazidime, cefuroxime, and amoxicillin (1 each). Other therapies known to prolong QT-time, were administrated in four patients, such as morphine (1), omeprazole (1), and oseltamivir (3).
Treatment and QTc group classification of the patients under study.
| Case nr | Treatment with HCQ (duration in days) | Treatment with LPV/r (n=3) | Treatment with Azithromycin (n=5) | Other cardiotoxic therapies | ECG – QTc time classificationa | ||
|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 96 h | |||||
| 1 | 5 | no | no | no | 1 | 1 | – |
| 2 | 5 | no | no | Omeprazole | 1 | 1 | 1 |
| 3 | 5 | no | no | no | 1 | 1 | 1 |
| 4 | 5 | no | no | no | 1 | 1 | 1 |
| 5 | 5 | no | no | no | 1 | 3 | 1 |
| 6 | 5 | no | yes | no | 2 | 1 | 1 |
| 7 | 7 | yes | no | no | – | – | – |
| 8 | 7 | yes | no | Oseltamivir | 1 | 1 | 1 |
| 9 | 5 | no | yes | Oseltamivir | 1 | 1 | 1 |
| 10 | 5 | no | yes | Ketamine+morphine | 1 | 3 | 2 |
| 11 | 5 | no | no | Oseltamivir | 1 | 1 | 2 |
| 12 | 5 | yes | no | no | 1 | – | 1 |
| 13 | 5 | no | yes | no | 2 | – | 1 |
| 14 | 5 | no | yes | no | 1 | – | 1 |
QTc time classification: Group 1 – QTc time normal for age; Group 2 – QTc time between normal values and 500 ms; Group 3 – QTc time the same or above 500 ms. When this value was not calculated it was described as “–”.CT – computed tomography; ECG: electrocardiogram; LPV/r: lopinavir/ritonavir, QTc: corrected QT time.
Baseline ECG was performed before the start of therapy in all patients, except in a seven month-old boy, as he was our first child treated with HCQ. According to their initial QTc, 11 patients were classified as Group 1 (normal QTc for age) and two children had values ranging from 460 to 470 ms, so they were classified as Group 2 (prolonged QTc but lower than 500 ms). Both of them were previously healthy (aged four and nine years) and were admitted for COVID-19 pneumonia. No change in blood ion levels (sodium, potassium, chlorine, calcium, magnesium and phosphorus) was noted. Coinfection with Mycoplasma pneumoniae was identified in both, and azithromycin was combined with HCQ.
Before implementation of the monitoring protocol, three children were already under HCQ treatment and QTc was only measured at admission and after 96 hours instead of the predefined point (48 hours). On the other hand, after 48 hours of therapy, QTc was again measured and calculated in 10 patients, and they were classified accordingly. Eight patients were defined as Group 1 from which seven had already been included in this group and one normalized his QTc. Moreover, two patients with a previously normal value were reclassified as Group 3 (QTc higher or the same as 500 ms) which led to temporary suspension of HCQ. Regarding these two cases, one child was a previously healthy 16-year-old girl. Her baseline QTc was 450 ms and she was treated in monotherapy with HCQ. After 48 hours, an increase of 50 ms in QTc to a value of 500 ms was recorded. Considering her clinical characteristics and normal blood electrolytes, therapy was suspended for 24 hours. A new ECG showed normalization of QTc, and HCQ was restarted. ECG performed at two, 24, and 48 hours showed normal QTc.
The other case was a 15-year-old boy with a history of sickle cell disease. When SARS-CoV-2 infection was confirmed, he had already been diagnosed with left elbow osteomyelitis and acute thoracic syndrome. He was treated with cefotaxime, clindamycin, morphine and ketamine. Pre-treatment QTc was 410 ms and he was started on HCQ and azithromycin. The ECG performed at 48 hours showed an increase to 510 ms. No ion changes were identified, and other therapies were revised but not suspended as they were considered essential for symptom control. HCQ was suspended for 24 hours, with a decrease of QTc to 480 ms. Therapy was reintroduced and ECG was performed, first, after two hours, and then, every 24 hours. QTc was always in normal range.
Finally, at 96 hours of therapy, 10 patients were reclassified as Group 1, and 2 children as Group 2. One of the mild disease group patients did not undergo the ECG as she was discharged home earlier. No other adverse effects of HCQ were reported during treatment.
DiscussionSince the beginning of COVID-19 pandemic, clinical trials have been developed in order to define the best therapy for the disease. Due to an immediate need for effective drugs, therapies already known to be active in other coronavirus, and for which security had already been proven in other clinical conditions, were proposed as experimental therapies for COVID-19. Lacking stronger scientific evidence, management and treatment protocols were based on case-reports and opinion expert papers from the first affected countries.19
Hydroxychloroquine was one of the proposed therapies for COVID-19. It belongs to the group of aminoquinolines which are approved for the prevention and treatment of malaria and several autoimmune diseases (as cutaneous and systemic lupus and rheumatoid arthritis).20 Both chloroquine and HCQ have been reported to inhibit SARS-CoV-2 in vitro,21 although HCQ appears to have more potent antiviral activity and fewer side effects than chloroquine. It appears to act by blocking the cellular entry of the virus through the inhibition of the receptor's glycosylation, proteolytic processing and endosomal acidification.22 It also seems to have an immunomodulatory effect by diminishing the production of pro-inflammatory cytokines and inhibiting the autophagic and lysosomal activity of the host's cells.22
Hydroxychloroquine was proposed as a treatment option for mild and moderate infection. Therefore, most guidelines started to include this therapy as part of treatment for COVID-19.13,23
Among its side effects, retinal toxicity is associated with prolonged exposure to the drug,24 however, the most alarming effect of its use in COVID-19 treatment is QT interval prolongation and the potentil risk of inducing life threatening arrythmias.25
As far as we know, this is the first study describing a population of pediatric patients with COVID-19 treated with HCQ and the use of a protocol for cardiac monitorization during therapy. Few pediatric series of COVID-19 patients have been published. Götzinger et al.26 published a European multinational, multicenter cohort study of pediatric patients with COVID-19. It included 582 patients, 40 of whom were treated with hydroxychloroquine.26 The median age of Götzinger's cohort was five years, while in ours the median was 10 years. We found a male to female gender ratio of 1:1.8, similar to the European cohort.26
Concerning severity, among the 14 patients, 10 were moderate or severe. Most studies on children reported a much lower incidence of severe disease.27,28 Some facts can partially explain this discrepancy. First, our study excluded mild patients treated at home with no specific therapy, only considering patient with criteria for hospital admission. Second, we report on the circumstances at one of the two tertiary pediatric hospitals named as referral hospital for management of COVID-19 cases in Portugal, meaning that patients diagnosed from other hospitals across the country were transferred to our unit depending on admission criteria, raising the number of severe patients. This highly selected population creates a bias, thus making it difficult to compare with other series.
The mean time between disease onset and diagnosis was six days, and HCQ was started with a delay of one day. Thus, most of our patients started therapy when the clinical syndrome was already stablished. Due the risk of decompensating pre-existing conditions, four patients with comorbidities were treated, even though only mild symptoms were noted.
Coinfection was found in six patients, with Mycoplasma pneumoniae, Chlamydia pneumoniae, adenovirus, metapneumovirus and rhinovirus. Similar results were observed by Wu et al.29 which found coinfection in 51% of pediatric COVID-19 patients, while other large studies found coinfection in about 5% of pediatric patients.26,28 This disparity can be justified in part by the difference in patient selection for analysis as there seems to be an association between coinfection and higher disease severity.26 This gains particular importance as some of these infections have a specific treatment path. Although patients with coinfection seem to be more prone to having respiratory symptoms at diagnosis, no clinical factors are specific to these children.26 Thus, considering the possibility, or identification of coinfection, almost all moderate and severe patients with pneumonia were also medicated with antibiotics, five of them with azithromycin.
Similar to HCQ, therapies such as lopinavir/ritonavir and azithromycin, as well as other overused therapies can also prolong QTc or interfere with their metabolism, enhancing cardiac adverse effects.30
QT interval prolongation has the potential for severe cardiac arrhythmias including Torsade de Pointes.31
Based on evidence from that time and in accordance with guidance from the Mayo Clinic,18 a protocol for QTc monitoring was adapted and applied to all pediatric patients admitted with COVID-19 and treated with HCQ. As the risk of cardiac arrhythmia with the use of some therapies is higher in patients with inherited arrhythmia syndromes,32 a baseline ECG was performed in all patients. In order to reduce drug interactions, the medical history was reassessed, and nonessential medicines were suspended, as were oral antihistamines (suspended in two patients). The baseline ECGs were normal in the majority of patients. Two children showed a normal registry, apart from a QTc slightly above normal value for their age, with normalization over time, and with no personal or family medical history of cardiac rhythm disturbances. The monitoring of QTc at 48 hours of treatment showed a prolongation to values above 500 ms in two patients. One of them was an otherwise heathy adolescent medicated with HCQ as monotherapy and with no other factors known to contribute to this effect. The other case was a male adolescent with a severe sickle cell disease phenotype, with acute decompensation (acute thoracic syndrome and osteomyelitis). Apart from the use of HCQ, he presented other risk factors for acquired long QT. Some studies have hypothesized an association between microvascular ischemia and QT prolongation with fatal arrhythmias in patients with sickle cell disease.33 Moreover, he was also medicated with other drugs known to prolong QTc which use had to be discussed, but we decided not to suspend them as they were essential for symptom and disease control.
Both of them temporarily suspended HCQ to prevent adverse effects. It reinforces the need for cardiac monitorization before and during treatment of COVID-19 in all patients, even in those with no other risk factors. It is also fundamental to review the concomitant therapy taken by all patients.
No other adverse effects from the therapy were reported.
The safety of drug combinations, especially in COVID-19 is not well studied. With the implementation of the cardiac monitoring protocol during treatment with HCQ, two patients experiened ECG changes with consequent therapy suspension. Therefore, it is essential to monitor cardiac rhythm during the use of HCQ to prevent concerning treatment complications.
Some studies have warned about the cardiac adverse effects of aminoquinolines. A Brazilian randomized clinical trial34 of chloroquine for adults with COVID-19 faced early suspension due to safety results. They found an association between higher doses of chloroquine and more severe adverse effects. Mortality was 39% in the group treated with high-dosage of the medicine versus a mortality of 15% in the low-dosage group, with a statistically significant value.34 Concomitantly, other studies in an adult population support the safety of HCQ and chloroquine. Saleh et al.35 studied a population of 201 COVID-19 patients >18 years old, treated with chloroquine or hydroxychloroquine, and their effect on QTc. Similar to our results on a pediatric population, they reported no episodes of Torsades de pointes associated with QTc prolongation or death due to arrythmia. However, 3.5% of patients had to suspend therapy due to QTc lengthening.35 He also found that the maximum QTc during treatment was significantly shorter in patients treated with monotherapy when compared with a combination with azithromycin.35 This result supports the idea that it is fundamental to consider the necessity of concomitant therapies when HCQ is going to be started.
Patients with some cardiac diseases are at an increased risk of drug-related proarrhythmic effect.36 Thus, it can be hypothesized that HCQ may have fewer adverse effects on children when compared with adults, as they may have lower cardiac lesion and fewer comorbidities. However, no safety studies of HCQ on COVID-19 have been done yet in children. Clinical trials are currently studying the effects of HCQ for treatment, pre- and post-exposure prophylaxis for COVID-19, as well as its safety.37,38
In June, WHO suspended the HCQ arm of the Solidarity Trial (a clinical trial designed to assess the effect and safety of four drugs on COVID-19), after The Lancet published a paper alerting to increased fatality rates when HCQ was used.39,40 Although the paper was later retracted, following the example of several other countries, HCQ was suspended from the Portuguese guidelines on treatment of COVID-19, pending new evidence.41
Only a few COVID-19 clinical trials currently taking place include the pediatric population, which compromises the development of knowledge on safe therapeutic options for children in the future. If new evidence shows benefits to the use of HCQ, we strongly believe that a protocol for cardiac adverse effect prevention and monitoring should be developed and promptly used for the whole of the treatment in order to prevent arrhythmias as an iatrogenic complication.
Our period of study is two months. Since HCQ use has been discouraged by most experts throughout the world, we do not expect to expand our experience in the near future. More robust studies, preferably clinical trials with a larger sample are needed, particularly in pediatric patients to confirm the safety of HCQ and the advantage of its use in COVID-19.
Finally, while new evidence may be enlightening us about the efficacy of HCQ in COVID-19, our study suggests that its use seems to be safe in the pediatric population, although it highlights the need for well-defined cardiac adverse effect monitoring during treatment.
ConclusionCardiac toxicity of SARS-CoV-2 and the relative contribution of concomitant drugs in the reported cardiac adverse effects in COVID-19 patients has not been well studied. This situation is particularly evident in the pediatric population where studies are not so common.
The discussion remains as to whether HCQ is our best weapon to treat COVID-19 patients or if the risk of cardiac events is greater than the benefit it brings. However, as the question remains, it is fundamental to monitor cardiac rhythm during treatment with HCQ in the pediatric population.
Conflicts of interestThe author has no conflicts of interest to declare.