Catheter ablation has been shown to improve left ventricular (LV) ejection fraction (LVEF) in patients with atrial fibrillation (AF) and heart failure (HF). Our aim was to assess the impact of AF ablation on the outcome of patients with HF and LV systolic dysfunction.
MethodsWe performed a retrospective observational cohort study of all patients with HF and LVEF <50% and with no apparent cause for systolic dysfunction other than AF who underwent catheter ablation in a tertiary referral center between July 2016 and November 2018. The primary endpoint was a ≥5% improvement in LVEF. Secondary endpoints included improvement in New York Heart Association (NYHA) class and reduction in LV end-diastolic diameter (LVEDD) and left atrial diameter (LAD).
ResultsOf 153 patients who underwent AF ablation in this period, 22 (77% male, median age 61 [IQR 54-64] years) fulfilled the inclusion criteria. Median follow-up was 11.1 months (IQR 6.1-19.0). After ablation, median LVEF increased from 40% (IQR 33-41) to 58% (IQR 55-62) (p<0.01), mean NYHA class improved from 2.35±0.49 to 1.3±0.47 (p<0.001), and median LAD and LVEDD decreased from 48.0 (IQR 43.5-51.5) mm to 44 (IQR 40-49) mm (p<0.01) and from 61.0 (IQR 54.0-64.8) mm to 55.0 (52.2-58.0) mm (p<0.01), respectively.
ConclusionIn patients with HF and LV systolic dysfunction, AF ablation is associated not only with improved functional status but also with favorable structural remodeling, including improvement in LVEF and decreases in LAD and LVEDD.
A ablação por cateter está associada a uma melhoria da fração de ejeção ventricular esquerda (FEVE) em doentes com insuficiência cardíaca (IC) e fibrilhação auricular(FA). O objetivo deste trabalho foi avaliar o impacto da ablação de FA em doentes com IC e disfunção sistólica do ventrículo esquerdo.
MétodosEstudo observacional retrospetivo dos doentes com IC e FEVE < 50% sem causa aparente para a disfunção ventricular para além da FA, submetidos a ablação num centro de referência entre julho/2016-novembro/2018. O endpoint primário foi uma melhoria da FEVE ≥ 5%. Os endpoints secundários foram a melhoria na classe funcional da New York Heart Association (NYHA) e a redução do diâmetro auricular esquerdo (DAE) e do diâmetro telediastólico do ventrículo esquerdo (DTDVE).
ResultadosDos 153 doentes submetidos a ablação de FA neste período, 22 foram incluídos (77% do género masculino, idade mediada de 61 anos [IQR 54-64]). O tempo de seguimento mediano foi de 11, 1meses (IQR 6,1-19,0). Após a ablação, a mediana da FEVE passou de 40% (IQR 33-41) para 58% (55-62), P < 0,01, enqanto a classe de NYHA passou de 2,35 ± 0,49 para 1,3 ± 0,47, P < 0,001, o DAE de 48,0 (IQR 43,5-51,5) mm para 44 (IQR 40-49)mm (P < 0,01) e o DTDVE de 61,0 (IQR 54,0-64,8)mm para 55,0 (52,2-58,0, P < 0,01).
ConclusãoEm doentes com IC e disfunção sistólica do ventrículo esquerdo, a ablação de FA associa-se a uma melhoria da classe funcional e a uma remodelagem estrutural favorável, incluindo melhoria da FEVE e diminuição do DAE e DTDVE.
Atrial fibrillation (AF) and heart failure (HF) are two major public health issues that often coexist. AF is responsible for a large number of cases of HF with reduced left ventricular (LV) ejection fraction (LVEF),1 and HF is associated with increased risk of AF. Several studies have demonstrated that HF symptoms may occur secondary to AF, and improve after restoration of normal sinus rhythm.1–3 However, studies on antiarrhythmic drug (AAD) therapy have failed to show a clear benefit for a rhythm control strategy, owing to the limited efficacy and potentially deleterious effects of such drugs.4 Importantly, in the context of HF, many AADs pose a significant proarrhythmic risk.5
Catheter ablation for AF has become a treatment option for patients with drug-refractory symptoms.6–9 Recent data have shown a favorable impact of catheter ablation in patients with AF and HF, reducing all-cause death and hospitalization.1,2,7,10–14
However, despite these encouraging data, the most recent European guidelines for HF management, as well as the 2019 clinical practice update, still attribute a weak level of recommendation for AF ablation in patients with HF with reduced ejection fraction (HFrEF) (class IIb recommendation, level of evidence B).5,15 By contrast, the guidelines for AF management state that AF ablation should be considered for symptomatic patients with HFrEF when tachycardia-induced cardiomyopathy is suspected (class IIa, level of evidence C).9
Given the differing levels of recommendation in the aforementioned guidelines, further studies are warranted. The aim of this study was to assess the impact of AF ablation on the outcome of patients with HF and LV systolic dysfunction.
MethodsStudy design and settingWe performed a retrospective observational cohort study of consecutive patients with HF symptoms and LVEF <50% who underwent AF catheter ablation between July 2016 and November 2018 in a tertiary referral center. Procedural endpoints and mid-term follow-up results were assessed.
Patient eligibility criteriaPatients were eligible for inclusion in the study if they met the following criteria: drug-refractory AF or intolerance to AAD therapy; LVEF <50%; and symptoms of HF (New York Heart Association [NYHA] functional class ≥2). All patients provided written informed consent prior to the procedure.
Paroxysmal AF was defined as AF lasting less than seven days, persistent AF as AF lasting between seven days and one year, and long-standing persistent AF as lasting for more than one year before a decision to attempt rhythm control was made.9
All patients underwent assessment for any underlying structural heart disease, and significant coronary artery disease had to be ruled out by cardiac angiography or stress testing.
Exclusion criteria were the following: age <18 years; severe valvular disease; ischemic cardiomyopathy (defined as a history of documented >75% left main or proximal left anterior descending artery stenosis, >75% two-vessel stenosis or a previous history of myocardial infarction and/or revascularization16 or a documented area of ischemia >10% of the left ventricle);5,17 hypertrophic cardiomyopathy; neuromuscular disease; congenital heart disease; other apparent cause for systolic dysfunction (apart from tachycardia-induced cardiomyopathy); and left atrial thrombus.
Data required for cohort characterization were collected from patients’ clinical records. Echocardiographic data including LVEF, left atrial (LA) diameter (LAD) and LV end-diastolic diameter (LVEDD) were also collected. These measurements were performed by specialized operators blinded to the study endpoints. LA anteroposterior diameter and LVEDD were measured at end-systole in M-mode images in parasternal long-axis views. LVEF was calculated according to current recommendations.18
Ablation procedureIn order to reduce time in AF, electrical cardioversion was performed in all patients with persistent AF before intervention. For this reason, a large number of patients were in sinus rhythm at the time of ablation. All procedures were conducted under general anesthesia and patients suspended AAD therapy at least five half-lives before the procedure, except for amiodarone.
All patients were under oral anticoagulation (OAC) for at least two months prior to the procedure. In patients taking vitamin K antagonists (VKAs), OAC was continued in the periprocedural period with an international normalized ratio within the 2.0-3.0 range. In patients taking non-VKA OAC (NOAC), the last NOAC dose before the procedure was omitted. The presence of intracardiac thrombus was excluded prior to the procedure by transesophageal echocardiography or cardiac computed tomography (CT). During catheter ablation, unfractionated heparin was administered immediately after transseptal puncture and adjusted as needed for a target activated clotting time >300 s.
Anatomical mapping data were collected using a three-dimensional mapping system – CARTO 3 (Biosense-Webster, Irvine, CA, USA) or NavX (Abbott, Chicago, IL, USA) – and integrated with a CT imaging reconstruction of the left atrium, when available.
Each patient underwent a tailored approach, according to our institutional protocol, irrespective of AF type (persistent or paroxysmal) and duration. Briefly, if patients were in sinus rhythm, bipolar voltage mapping was performed. LA mapping was considered to be adequate when >500 voltage points were acquired. Voltage criteria for healthy and scarred tissue were >0.5 mV and <0.2 mV, respectively. After LA mapping, point-by-point pulmonary vein isolation (PVI) was carried out.
If the patient was in AF at the beginning of the procedure, PVI was performed first. Electrical cardioversion was subsequently performed if patients remained in AF after PVI, and a voltage map was obtained in sinus rhythm.
In the presence of low voltage areas, if the patient remained in AF or AF remained inducible with atrial burst pacing, these areas were ablated to achieve tissue homogenization, and if necessary lines were created connecting areas of scar to previous ablation lines (with the aim of achieving bidirectional block). In the absence of low voltage areas but persisting AF inducibility, ablation of complex fractionated atrial electrograms (CFAEs) was performed.19 If AF was converted to a regular atrial arrhythmia, this was mapped and ablated. Likewise, in the presence of typical flutter, ablation of the cavotricuspid isthmus was also performed. When AF persisted or was inducible at the end of the procedure, electrical cardioversion was performed.
Study endpointsThe primary endpoint was improvement in LVEF, of at least 5% in order to reduce inter-operator variability.20
Secondary endpoints included improvement in NYHA class and reductions in LVEDD and maximum anteroposterior LAD. AF recurrence after catheter ablation was also analyzed in this population subset.
Safety endpoints included any major procedure-related complications (death, cardiac tamponade, stroke or systemic embolism, or vascular injury requiring transfusion or intervention).
Follow-upPatients were assessed before discharge and at three, six and 12 months after the procedure. Transthoracic echocardiography (TTE) and 24-hour Holter monitoring were performed before discharge. Information collected during follow-up included clinical data, 24-hour or seven-day Holter monitoring at each visit, and TTE at six or 12 months. Recurrence of AF was defined as the documentation of at least 30 s of AF, atrial tachycardia or atrial flutter, irrespective of symptoms, in accordance with the Expert Consensus Statement on Catheter and Surgical Ablation of Atrial Fibrillation.21
No AADs were prescribed after ablation in paroxysmal AF patients. In those with persistent AF, AADs were prescribed on discharge at the discretion of the operator (particularly in patients with low voltage areas or persistence of AF at the end of the procedure requiring electrical cardioversion). The first three months after the procedure were considered the blanking period. The anticoagulation strategy after the first three months was based on the CHA2DS2-VASc and HAS-BLED scores.9
Statistical analysisStatistical analysis was performed using IBM SPSS Statistics version 20 (IBM, Armonk, NY, USA). Categorical variables are expressed as frequencies and percentages and continuous variables as median and interquartile range (IQR), for variables with or without a normal distribution, respectively. The chi-square test was used to assess differences between categorical variables and the Student's t test or the Wilcoxon test were used to compare continuous variables with or without a normal distribution, respectively. The Kolmogorov-Smirnov test was used to test for normality of distribution of continuous variables. Statistical significance was accepted for p-values <0.05.
ResultsDuring the enrollment period, a total of 153 patients underwent AF ablation. Of these, 22 (14.4%) patients fulfilled the inclusion criteria and were included in the study (77% male, median age 61 [IQR 54-64] years).
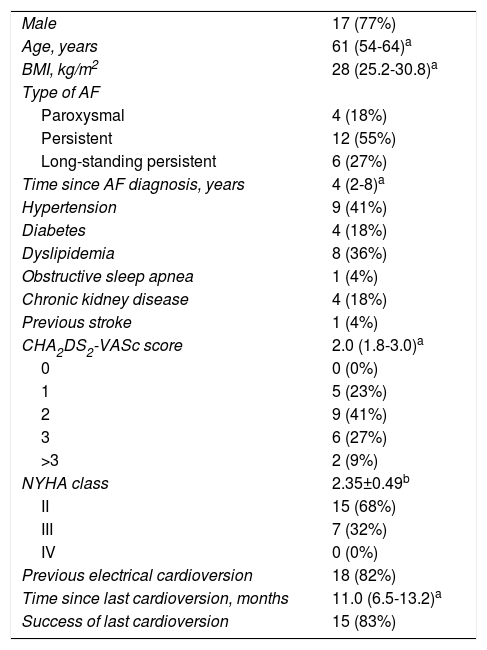
The main baseline population characteristics are detailed in Table 1. The majority of patients (82%) presented with persistent AF (including 27% with long-standing persistent AF), while 18% had paroxysmal AF (all patients with paroxysmal AF underwent electrical cardioversion in the first seven days of hospitalization, when the diagnosis was initially made). The median heart rate was 93 (IQR 84-102) bpm. Regarding NYHA class, 32% of patients were in class III, while the remainder were in NYHA class II. At baseline, medical treatment was reasonably optimized. Importantly, all patients with LVEF <40% were under renin-angiotensin system inhibitors plus beta-blocker therapy, and 56% were also taking an aldosterone antagonist, while 77% of patients (17) were under antiarrhythmic therapy at baseline, mostly (82%, 14 patients) amiodarone.
Baseline characteristics of the study population (n=22).
| Male | 17 (77%) |
| Age, years | 61 (54-64)a |
| BMI, kg/m2 | 28 (25.2-30.8)a |
| Type of AF | |
| Paroxysmal | 4 (18%) |
| Persistent | 12 (55%) |
| Long-standing persistent | 6 (27%) |
| Time since AF diagnosis, years | 4 (2-8)a |
| Hypertension | 9 (41%) |
| Diabetes | 4 (18%) |
| Dyslipidemia | 8 (36%) |
| Obstructive sleep apnea | 1 (4%) |
| Chronic kidney disease | 4 (18%) |
| Previous stroke | 1 (4%) |
| CHA2DS2-VASc score | 2.0 (1.8-3.0)a |
| 0 | 0 (0%) |
| 1 | 5 (23%) |
| 2 | 9 (41%) |
| 3 | 6 (27%) |
| >3 | 2 (9%) |
| NYHA class | 2.35±0.49b |
| II | 15 (68%) |
| III | 7 (32%) |
| IV | 0 (0%) |
| Previous electrical cardioversion | 18 (82%) |
| Time since last cardioversion, months | 11.0 (6.5-13.2)a |
| Success of last cardioversion | 15 (83%) |
a Median and interquartile range.
b Mean ± standard deviation.
AF: atrial fibrillation; BMI: body mass index; IQR: interquartile range; LVEF: left ventricle ejection fraction; NYHA: New York Heart Association.
Median LVEF was 40% (IQR 34.5-40.2); 41% of the patients had HFrEF (i.e. LVEF ≤40%) and the remainder had HF with mid-range ejection fraction (HFmrEF), as defined by the 2016 European guidelines.5 Median LAD was 48.0 (IQR 43.5-51.5) mm and median LVEDD was 51.0 mm (IQR 54.0-64.8). Data regarding baseline echocardiographic parameters are presented in Table 2.
Baseline echocardiographic parameters (n=22).
| LVEF, % | 40.0 (IQR 34.5-40.2) |
| LVEF ≤35% | 7 (32%) |
| LAD, mm | 48.0 (IQR 43.5-51.5) |
| LVEDD, mm | 61 (IQR 54.0-64.8) |
IQR: interquartile range; LAD: left atrial diameter; LVEDD: left ventricular end-diastolic diameter, LVEF: left ventricular ejection fraction.
Details of the technical aspects of the procedures are presented in Table 3. A significant proportion of patients (54%) were in sinus rhythm at the beginning of the procedure, reflecting the high percentage of patients who had undergone electrical cardioversion in the preceding months. All patients underwent a single procedure. A voltage map was acquired in sinus rhythm in all patients, and low-voltage areas outside the pulmonary veins were detected in seven (32%) and CFAEs in five (23%). Antral PVI was performed successfully in all patients, with bidirectional block being obtained at first passage in 73% of cases. Low-voltage homogenization and lines were performed in six (27%) patients, CFAE ablation in five (23%) and cavotricuspid isthmus line ablation in eight (36%). At the end of the procedure, AF was still inducible in four patients (18%), and these underwent successful electrical cardioversion.
Procedural details.
| SR at beginning of procedure | 12 (55%) |
| AF at beginning of procedure | 10 (45%) |
| Procedural time, min | 116±33 |
| Ablation time, min | 40±14 |
| Fluoroscopy time, min | 5.3±3.0 |
| Voltage map | 22 (100%) |
| Low voltage area (outside PVI lines) | 7 (32%) |
| CFAE ablation | 5 (23%) |
| PVI at first passage | 18 (82%) |
| Low voltage/empirical lines | 6 (27%) |
| CTI line | 8 (36%) |
AF: atrial fibrillation; CFAE: complex fractionated atrial electrogram; CTI: cavotricuspid isthmus; PVI: pulmonary vein isolation; SR: sinus rhythm.
Continuous variables are expressed as mean ± standard deviation.
There were no immediate procedural or in-hospital complications.
Study endpointsMedian follow-up was 11.1 (IQR 6.1-19.0) months.
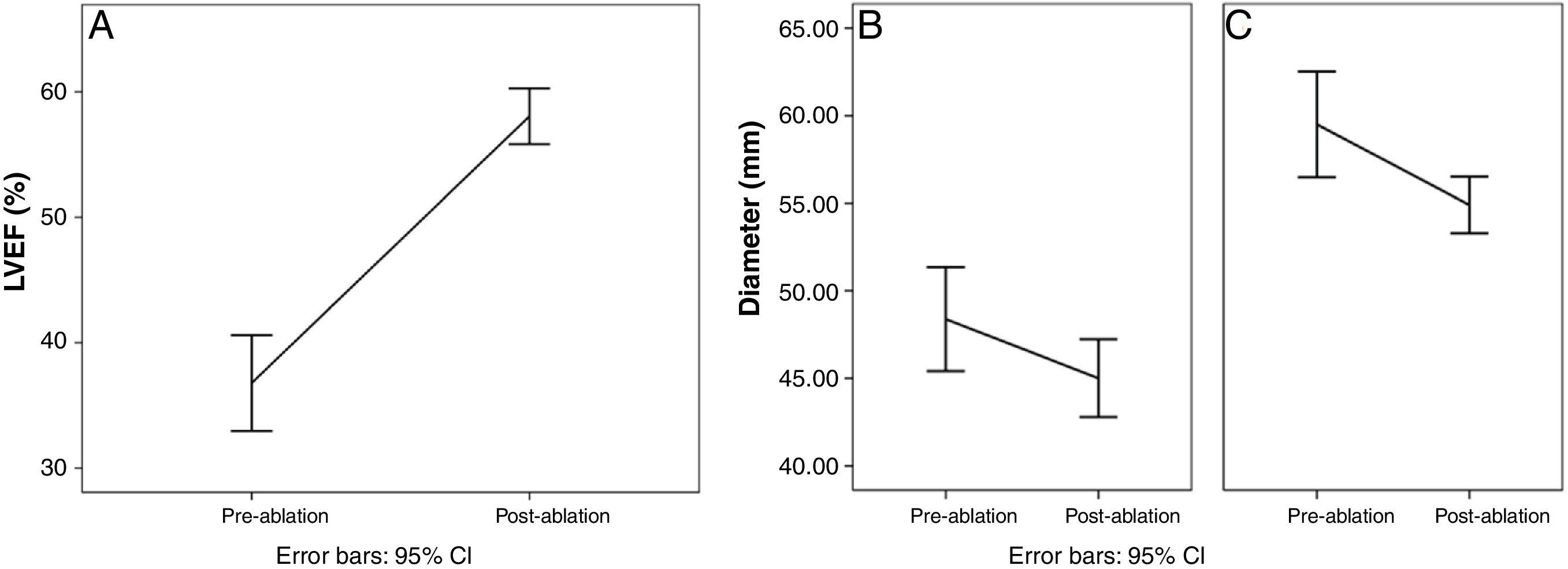
LVEF assessment after catheter ablation was available in all cases. Improvement was observed from 40% (IQR 33-41) to 58% (IQR 55-62) (median 20% [IQR 15-26], p<0.01). Changes in LVEF before and after the procedure are shown in Figure 1A. Importantly, in 21 cases (95%), LVEF after catheter ablation was >50%. In fact, improvement in LVEF was observed even in patients with AF recurrence. Of the four patients with AF recurrence documented during follow-up, only one still had LVEF <50% (this patient had a considerable improvement from 16% to 43%), while in the remainder LVEF improved to >50%.
LAD and LVEDD measurements were available in 21 (95%) and 20 (91%) patients, respectively. LAD decreased from 48.0 (IQR 43.5-51.5) mm to 44 (IQR 40-49) mm (p<0.01), while LVEDD decreased from 61.0 (IQR 54.0-64.8) mm to 55.0 (52.2-58.0) mm (p<0.01). Changes in LAD and LVEDD are shown in Figure 1B and C.
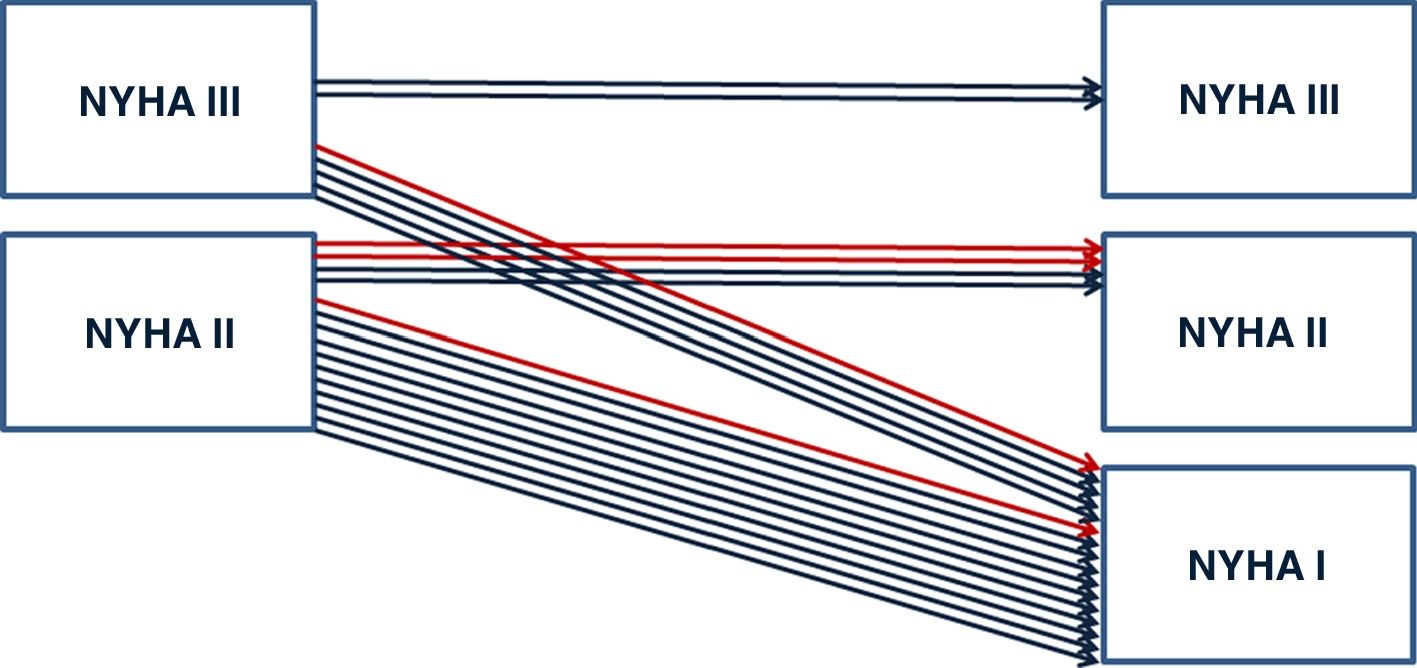
Mean NYHA class improved from 2.35±0.49 to 1.3±0.47 (p<0.01) after catheter ablation. Figure 2 shows improvements in NYHA class pre- and post-ablation. At last follow-up, diuretic doses were reduced or discontinued in 55% of the patients.
Recurrence after the blanking period was documented in 18% of patients, four in total – three in AF (two of whom were in paroxysmal AF) and one in paroxysmal atrial tachycardia; one patient was under AAD therapy at the time of recurrence.
All recurrences were in patients with a history of persistent AF (three with long-standing persistent AF), meaning that freedom from symptomatic and/or documented AF at last follow-up was 78% for patients with persistent AF and 100% for those with paroxysmal AF with a single ablation procedure. Only two patients remained under amiodarone at follow-up.
There were no major procedure-related complications. No patients died during follow-up and there were no hospital admissions due to cardiovascular causes.
DiscussionThe main findings of this study are that AF catheter ablation is associated with significant improvements in both LVEF and NYHA functional class in patients with HF and LV systolic dysfunction, and that AF catheter ablation induces favorable atrial and ventricular reverse remodeling in this patient subset.
Although there have been several randomized clinical trials assessing the role of AF catheter ablation in patients with LV systolic dysfunction, evidence for significant improvement of LVEF post-ablation is scarce, with some studies even reporting a lack of improvement,7,22–24 which may have been the result of less selective inclusion criteria, with sicker patients and less favorable HF etiologies. By contrast, in our study we observed significant improvements in LVEF (from a median of 40% [IQR 33-41] to 58% [IQR 55-62], p<0.01) and all patients showed at least partial LVEF improvement. We further observed significant reverse remodeling after catheter ablation, with reductions in LA diameter and LVEDD. These results are in line with studies which included patients with idiopathic cardiomyopathy.1,2,14,25–27 Overall, the reductions in cardiac chamber dimensions and recovery of LV systolic function after ablation support the theory that, in the absence of additional underlying cardiac disease, reverse remodeling takes place once the primary insult (AF) is resolved. Also, these data suggest that patient selection plays a major role in defining the benefit driven by ablation and that patients with truly idiopathic cardiomyopathy or suspected tachycardia-induced cardiomyopathy benefit the most from a catheter ablation-based rhythm control approach.28,29 We should, however, acknowledge that there are other mechanisms by which AF may lead to systolic impairment, including irregular ventricular activity, loss of atrial contraction and even neurohormonal aspects,2 and the benefit of ablation may also result from the correction of these mechanisms.
Our study showed improvement in NYHA functional class after ablation in patients with HF and LV systolic dysfunction, with reduction or discontinuation of diuretics possible in over half of the patients. It is also noteworthy that even patients with AF recurrence still saw improvements in functional class and LVEF, which may be explained by the significant reduction in AF burden.1,6 These encouraging results were observed on top of appropriate medical therapy. Importantly, as in the recent Catheter Ablation versus Standard Conventional Therapy in Patients with Left Ventricular Dysfunction and Atrial Fibrillation (CASTLE-AF) trial,10 most of our patients were in NYHA class II, and these results should therefore not be extrapolated to patients with more severe forms of HF. Nevertheless, it would seem reasonable to expect improvement in these patients as well, even if of lesser magnitude, provided that other underlying heart conditions have been excluded.
In our study, 18% of patients had AF recurrence after the blanking period, all with persistent AF and 75% off AAD therapy, which is in line with some reports.7,14,26,30 However, 52% of patients continued AAD therapy during follow-up (despite a reduction in amiodarone from 64% at baseline to 9% in follow-up). This high percentage of patients on AADs after almost a year of follow-up derives mainly from concern that potential AF recurrence could lead to recurrent HF symptoms and deterioration of LV systolic function. Nevertheless, our study suggests that AF ablation can be carried out with high efficacy in HF patients with significant LV systolic dysfunction. Therefore, since catheter ablation is the most effective treatment for maintenance of sinus rhythm and reduction of AF burden, it is logical that a strategy of ablation may provide substantial benefit in HF patients compared with medical treatment alone. This had already been suggested by important trials such as CASTLE-AF, AATAC and a sub-group analysis of CABANA, and is further corroborated by the results of our analysis.7,10,31
According to the most recent guidelines,5 32% of our patients would qualify for a device for primary prevention of sudden cardiac death. However, catheter ablation was followed by significant atrial and ventricular remodeling, which led to LVEF improvement in most cases. At one-year follow-up, no patient still fulfilled criteria for ICD implantation, irrespective of AF recurrence. This is of particular importance if we consider that these patients are usually relatively young1 (median age 60±8 years in our study) and would therefore have a high likelihood of device-related complications during their lifetime.32–34 Catheter ablation has been shown to be superior to atrioventricular node ablation with biventricular pacing in patients with HF.23 For patients with HF symptoms and baseline left bundle branch block, who would otherwise be potential candidates for cardiac resynchronization therapy (CRT), AF ablation may in fact lead to recovery of LV function to the point that CRT may no longer be indicated. Care should be taken, however, to ensure that these patients do recover post-ablation, considering the unequivocal benefit of CRT in those in whom significant LV systolic dysfunction persists. Still, the present findings, together with data from CASTLE-AF10 and the AATAC trial,7 suggest that catheter ablation in AF patients with suspected tachycardia-induced cardiomyopathy may obviate the need for implantable cardioverter-defibrillators and/or CRT devices and should perhaps be considered as first-line therapy for patients with AF and HF with systolic impairment that is seen as being directly caused by AF.
Study limitationsWe acknowledge several limitation of our work. First, this was a retrospective study involving a small number of patients treated in a single center without a control group, which precludes an objective assessment of the impact of ablation compared with medical treatment alone. Second, since our patients did not undergo cardiac magnetic resonance imaging prior to AF ablation, we could not differentiate between patients with tachycardia-induced cardiomyopathy and those with dilated cardiomyopathy due to other causes. However, it may still be possible to reverse LV dysfunction to some extent in the latter group. Third, our follow-up duration was relatively limited and we are thus unable to comment on the benefit of ablation in the mid to long term. Fourth, systematic monitoring using an implantable loop recorder could have documented a higher rate of asymptomatic AF recurrence and provided more objective information regarding AF burden in follow-up. Lastly, electrical cardioversion performed pre-ablation may have contributed to the improvements in functional class and LVEF, as well as atrial and ventricular remodeling. However, even on AADs, almost half of our patients had AF recurrence and were in AF at the beginning of the procedure, which strengthened the indication for it and clearly demonstrates that a similar level of benefit would not have been achieved with electrical cardioversion plus AAD therapy alone.
ConclusionIn carefully selected patients with AF and HF with LV systolic dysfunction, catheter ablation results in significant improvement in LV ejection fraction and decreases in LV and atrial diameters, as well as improvement in functional status. Consideration should be given to performing ablation at earlier stages in these patients, rather than adopting a more conservative strategy with antiarrhythmic drug therapy.
Ethical statementAll patients provided written informed consent and the study was approved by the local institutional ethics committee.
FundingThe authors received no funding for this work.
Conflicts of interestThe authors have no conflicts of interest to declare.