This is the first study in Angola with the aim of characterizing ventricular septal defect (VSD) among children and adolescents.
MethodsA cross-sectional study based on echocardiographic records of the largest pediatric cardiology center in Angola included all children and adolescents (0 to 18 years old) with VSD between April 2010 and March 2011. The diagnosis was made by transthoracic and Doppler echocardiography with a Medison SA 8000 system. The sample was divided into two groups: Group 1, isolated VSD; and Group 2, VSD associated with other congenital heart defects (CHDs). Age, gender, type of VSD, associated CHDs and genetic syndromes were assessed.
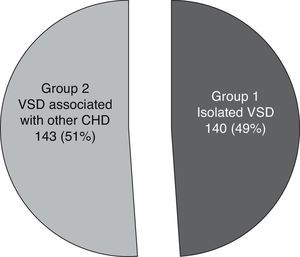
ResultsA total of 490 CHDs were diagnosed, of which 283 were VSDs. In Group 1 (140, 49%) the mean age was 29±36 months. The most frequent age (mode) at diagnosis was 24 months. There was no predominance of gender (ratio 1:1). The majority (127, 91%) had perimembranous VSD. In Group 2 (143, 51%) 113 patients (79%) had one, 27 patients (19%) had two and three patients (2%) had three other CHDs. Trisomy 21 was the most common genetic syndrome (23, 96%).
ConclusionsThe study shows that VSD is the most common CHD in childhood, the diagnosis is made late and almost half of VSDs are associated with other CHDs.
Esta é a primeira pesquisa em Angola com o objetivo de caracterizar a comunicação interventricular entre crianças e adolescentes.
MétodosEstudo transversal, com base nos registos ecocardigráficos do maior Hospital de Cardiologia Pediátrica em Angola (abril de 2010 para março de 2011). Todas as crianças e adolescentes (0 a 18 anos de idade) com comunicação interventricular foram incluídas. O diagnóstico foi feito por ecocardiografia transtorácica e Doppler com a máquina de Medison SA 8000. A amostra foi dividida em dois grupos: Grupo I – comunicação interventricular isolada e Grupo II – comunicação interventricular associada a outras cardiopatias congénitas. A Idade, o sexo, o tipo de defeito do septo, principais cardiopatias congénitas associadas e as síndromes genéticas foram avaliados.
ResultadosHouve 490 cardiopatias congénitas diagnosticadas, das quais 283 foram comunicação interventricular. No Grupo I (140, 49%) a idade média foi de 29±36 meses. A idade mais frequente (moda) ao diagnóstico foi de 24 meses. Não houve predomínio quanto ao género (1: 1). Neste grupo a maioria tinha comunicação interventricular perimembranosa (127, 91%). No Grupo II (143, 51%) 113 pacientes (79%) tinham uma, 27 (19%) tinham duas e três (2%) tinham três outras cardiopatias congénitas associadas a comunicação interventricular. A Trissomia do cromossomo 21 foi a síndrome genética mais comum (24, 96%).
ConclusõesO estudo mostra que comunicação interventricular é a cardiopatia congénita mais comum em crianças pré-escolares.
Ventricular septal defect (VSD) is the most common congenital heart defect (CHD), affecting approximately 2–6/1000 live births.1,2 VSD results from a malfunction of cardiac development, possibly including a failure in the alignment or in the fusion of the atrioventricular cushions during the formation of the interventricular septum.3 The defects can be classified according to their location. Perimembranous defects are located in the membranous portion of the interventricular septum and muscular defects are located in the muscular portion of the trabecular septum or at its margins.3 Perimembranous defect is more common, occurring in 70–80% of cases.4,5 VSDs can present in isolation or coexisting with other CHDs.
In Africa VSD is also the most common CHD in childhood.6–9 In Angola, little is known of the prevalence of VSD, largely because few studies have been conducted.10 The authors of this study aim to characterize VSD in children and adolescents based on echocardiographic records from the pediatric cardiology service of the largest pediatric care center in Angola over a period of one year, from April 2010 to March 2011.
MethodsThe David Bernardino Pediatric Hospital is the largest pediatric cardiology and care center in Angola. It is a tertiary teaching and postgraduate hospital with 300 beds in 11 departments. The cardiology service has an echocardiography laboratory with teleconsultation links to the pediatric cardiology department of Coimbra Hospital in Portugal. Around the country many pediatrics patients have been referred to the hospital for assessment. Consecutive echocardiographic reports of 1267 patients over a period of one year (from April 2010 to March 2011) were retrospectively reviewed. Repeat scans were not included. All children and adolescents aged 0–18 years with a diagnosis of VSD with or without associated CHD were included. The diagnosis of VSD was made by clinical examination and confirmed by transthoracic echocardiography performed by three cardiologists of the institution. The echocardiogram was performed on a Medison SA 8000 system, using an F24AC transducer.
The sample was divided into two groups, Group 1, isolated VSD; and Group 2, VSD associated with other CHDs.
The variables studied were age (months), gender, type of VSD, other CHDs and associated genetic syndromes. The data were presented as absolute and relative frequencies.
Descriptive statistics was used to determine mean, mode and standard deviation. The software used was Epi Info version 3.5.2.
ResultsOver a period of a year (April 2010–March 2011), 490 CHDs were diagnosed in children and adolescents (0–18 years of age), of which 283 (57.7%) were VSDs. Figure 1 shows the division of the sample into groups: Group 1, isolated VSD (140; 49%); and Group 2, VSD associated with other CHDs (143; 51%). In Group 1, the mean age at diagnosis was 29 months (SD 36 months). The most frequent age (mode) at diagnosis was 24 months. There was no predominance of gender (ratio 1:1). The most common type of VSD was perimembranous (127; 91%) with 13 cases of muscular VSD (9%). In this group, five children (3.6%) had pulmonary hypertension. In Group 2, 113 (79%) had one, 27 (19%) had two and three (2%) had three other CHD associated with VSD (Table 1). In this group, 24 (17%) had VSD associated with a genetic syndrome; of these, 23 (96%) had trisomy 21 and one (4%) had Noonan syndrome (Table 2). In Group 2, 50 (35%) of VSDs were associated with patent ductus arteriosus, 24 (16.8%) with pulmonary stenosis, eight (8.4%) with ostium primum atrial septal defect and seven (7.7%) with ostium secundum atrial septal defect (Table 3).
Prevalence of other congenital heart defects in patients with ventricular septal defect.
| Type of CHD | n (%) |
| PDA | 50 (35) |
| PS | 24 (16.8) |
| OSASD | 12 (8.4) |
| OPASD | 11 (7.7) |
| OSASD + PDA | 8 (5.6) |
| OPASD + PDA | 7 (4.9) |
| TGV | 3 (2.1) |
| CoA | 3 (2.1) |
| PDA + DORV | 3 (2.1) |
| TA | 2 (1.4) |
| CAVD | 2 (1.4) |
| DORV | 2 (1.4) |
| PCA + PS | 2 (1.4) |
| DORV + OSASD | 2 (1.4) |
| TOF | 2 (1.4) |
| AS | 1 (0.7) |
| Truncus arteriosus | 1 (0.7) |
| PDA + TGV + PS | 1 (0.7) |
| TGV + OPASD + PDA | 1 (0.7) |
| OPASD + PDA + PS | 1 (0.7) |
| PE + TA | 1 (0.7) |
| OPASD + TGV | 1 (0.7) |
| PS + DORV | 1 (0.7) |
| PS + AS | 1 (0.7) |
| OPASD + PDA | 1 (0.7) |
| Total | 143 (100) |
AS: aortic stenosis; CAVD: common atrioventricular canal defect; CHD: congenital heart defect; CoA: coarctation of the aorta; DORV: double outlet right ventricle; OPASD: ostium primum atrial septal defect; OSASD: ostium secundum atrial septal defect; PA: pulmonary atresia; PDA: patent ductus arteriosus; PS: pulmonary stenosis; TA: tricuspid atresia; TGV: transposition of the great vessels; TOF: tetralogy of Fallot.
In the present study the incidence of CHDs was higher than is found in the literature, probably due to the fact that this center receives children and adolescents from all over the country. VSD was the most common CHD, as it is worldwide.4–13 The prevalence of VSDs found in this study was also high compared to published reports.6–14 More than half of VSDs were associated with other CHDs, which contrasts with the results of some authors.9,12 This difference may be explained by the exclusion of patients with conotruncal anomalies, atrioventricular septal defect and single ventricle. On the other hand, our results were similar to those of a study at Hospital Infantil Pequeno Principe in Brazil.13 In cases of isolated VSD there was no gender predominance (ratio of 1:1), contrasting with the results of a study conducted in Enugu, Nigeria in which there was a predominance of males. The authors claimed this was due to Nigerian sociocultural practices.9 The age range of patients at diagnosis was wide (between six days and 18 years), which reflects the lack of a screening program and hence early diagnosis. Most VSDs were diagnosed at the age of 24 months (mode), which places Angola within the typical range of most developing countries.6–8 This result contrasts with those reported by other authors, who emphasize the need for diagnosis in the first year of life.9,12,13 Delay in diagnosis is directly proportional to a worse prognosis. Generally it is in the first year of life that the clinical manifestations of heart disease with left-to-right shunting appear, as in VSD, due to decreased pulmonary vascular resistance after birth and increased systemic resistance and right ventricular compliance, which causes the left-to-right shunt, leading to pulmonary hypertension and delay in development. The most frequent type of VSD in the isolated VSD group was perimembranous, which coincides with the results of other authors,4,14 but differs from those in a study at Cathay General Hospital (Taiwan) in which the authors found that 65% of VSDs were muscular.15 This may be an Asian characteristic. The percentage of association of VSD with other CHDs observed in this study is generally similar to the results of Glen et al.,12 although the association of VSD with two or three other CHDs was twice as common as in the literature. Regarding the type of CHD most commonly associated with VSD, our results differed from those of another study12 in which the most frequent associated CHD was pulmonary infundibular pulmonary stenosis, followed by aortic valve prolapse, pulmonary valve stenosis, ostium secundum atrial septal defect, patent ductus arteriosus and coarctation of the aorta. However, they resemble those of another African study (in Nigeria).9 As in our series, in another study trisomy 21 was the most frequent genetic syndrome (3.5%).12
ConclusionThe study demonstrated that VSD is the most common CHD in childhood. Delayed diagnosis is associated with worse prognosis. When VSD is detected we consider a detailed Doppler echocardiographic examination is required to exclude other CHDs associated with the VSD.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of interestThe author has no conflicts of interest to declare.
We would like to thank Feliciano Chanana Paquissi for his valuable contribution.