There is a marked contrast between the high prevalence of hypertension and the low rates of adequate control. A subset of patients with suboptimal blood pressure control have drug-resistant hypertension, in the pathophysiology of which chronic sympathetic hyperactivation is significantly involved. Sympathetic renal denervation has recently emerged as a device-based treatment for resistant hypertension. In this review, the pathophysiological mechanisms linking the sympathetic nervous system and cardiovascular disease are reviewed, focusing on resistant hypertension and the role of sympathetic renal denervation. An update on experimental and clinical results is provided, along with potential future indications for this device-based technique in other cardiovascular diseases.
A elevada prevalência da hipertensão está em claro contraste com a sua ainda insuficiente taxa de controlo. Um importante subgrupo destes doentes apresenta uma hipertensão resistente aos fármacos, na qual a hiperativação crónica do sistema nervoso simpático tem importantes implicações fisiopatológicas. Recentemente, a desnervação simpática renal emergiu como um tratamento de intervenção para a hipertensão arterial resistente. No presente artigo, são revistos os mecanismos fisiopatológicos subjacentes à interação entre o sistema nervoso simpático e as doenças cardiovasculares, com particular enfâse na hipertensão arterial resistente e no papel da desnervação simpática renal. É igualmente feita uma atualização dos resultados de estudos experimentais e clínicos, bem como de potenciais futuras indicações desta técnica de intervenção noutras doenças do foro cardiovascular.
blood pressure
confidence interval
estimated glomerular filtration rate
sympathetic nervous system
renin-angiotensin-aldosterone system
renal denervation
radiofrequency
Hypertension is the leading global risk factor for cardiovascular mortality, accounting for more than nine million deaths worldwide in 2010.1 Its close association with myocardial infarction, heart failure, stroke, end-stage renal disease and cardiovascular death is well established, with 54% of stroke and 47% of ischemic heart disease worldwide attributable to high blood pressure (BP).2 Effective BP lowering has consistently been shown to reduce overall cardiovascular risk,3 but rates of adequate BP control remain suboptimal, despite the wide range of antihypertensive drugs available and strong evidence supporting their use. A recently published study confirmed that rates of BP control in European countries are low, with only 37% of treated hypertensive patients achieving recommended BP values.4
The blame for such low rates cannot be attributed only to poor treatment. Resistant hypertension has a prevalence ranging from 15% to 30% of treated hypertensive patients,5 and is an important cause of failure of BP control. Most importantly, these patients exhibit a worse prognosis, with a higher risk for cardiovascular events, compared to hypertensive patients without resistant hypertension.6
In recent decades, the renin-angiotensin-aldosterone system (RAAS) has been the central focus of hypertension treatment and management. The availability of safe, effective and evidence-based drugs that block this system has meant that the role of other systems, particularly the autonomic nervous system, has been neglected.
The sympathetic nervous system (SNS) and its possible role in the pathogenesis of hypertension is receiving increasing attention. The aim of this review is to provide an update on the current understanding of the role of the SNS in blood pressure control and its implications for sympathetic renal denervation (RDN).
The sympathetic nervous system and cardiovascular diseaseThe development of open surgical sympathectomy in the 1930s highlighted the role of the SNS in severe hypertension, since it appeared to be effective in lowering high BP in patients with severe hypertension.7,8 However, the procedure was abandoned due to its poorly tolerated side effects and high surgical risk, especially after the appearance of ganglionic blockers, the first effective antihypertensive drug class.9
The recent development of a new device-based approach to treat severe resistant hypertension, through RDN, focused attention on the already well-known role of the SNS in initiating and maintaining high BP in patients with essential hypertension.10,11
Assessment of the sympathetic nervous system in humansThe major reason that the SNS has been so neglected is not because there are doubts concerning its critical role in the pathogenesis of hypertension and other cardiovascular diseases, but because it has been difficult to study and test this relation, due to the complex and clinically impractical methods used for assessing the SNS in humans. Until the early 1970s, the most common techniques were measurements of blood levels and urine excretion rates of norepinephrine and its derivatives, which provide a gross estimate of whole-body sympathetic activity at best.12 Since then, new methods have emerged for measuring sympathetic nerve firing rates in subcutaneous nerves and for assaying plasma concentrations of sympathetic transmitters.
Microneurography, a technique reported first by Hagbarth and Vallbo,13 provided a tool to study nerve firing in subcutaneous sympathetic nerves in skin and skeletal muscle vessels. It is based on recording bursts of nerve activity, synchronous with the heartbeat, generated in skeletal muscle vascular efferent nerves, through tungsten electrodes inserted in the skin. It is highly reproducible and closely related to sympathetic traffic directed to other structures and can be repeated over time, allowing assessment of the effects of interventions, direct quantification of sympathetic nerve traffic regulating vasomotor tone, and study of instantaneous reactions to rapid stimuli.
The spillover technique for measurement of norepinephrine release, first applied by Esler et al,14 is an isotope dilution method that calculates the clearance and spillover of norepinephrine, using an intravenous infusion of tritium-labeled norepinephrine. The relationship between the sympathetic nerve fiber firing rate of an organ and the rate of norepinephrine spillover into the venous effluent of that organ provides the rationale for using measures of regional norepinephrine release as a surrogate for sympathetic tone in individual organs,15 enabling assessment of regional sympathetic nervous function in humans.
This technique was central to the demonstration that heart failure patients had sympathetic overactivity rather than sympathetic denervation, as thought at the time, and opened the way to the routine use of beta-blockers in heart failure.16
Sympathetic nervous system overactivityBesides its central role in cardiovascular homeostasis, by controlling vascular tone through vasoconstriction of small resistance arteries, the sympathetic system also affects and regulates numerous other physiological processes (Table 1). There is growing evidence that sustained chronic changes in sympathetic activity are involved in the pathogenesis of many disease states, from metabolic to psychological disorders, including ischemic heart disease,17 chronic heart failure,18,19 hypertension,20–22 kidney disease,23 type 2 diabetes,24 obesity,24 metabolic syndrome,24 obstructive sleep apnea,25 depression26 and inflammatory bowel disease.27 A chronically overactive sympathetic system is linked to a worse prognosis in patients with heart failure and end-stage renal disease.28,29
Effects of increased sympathetic nerve activity.
| Vascular |
| Smooth muscle cell hypertrophy and proliferation |
| Endothelial dysfunction and damage |
| Arterial stiffness |
| Impairment of postural blood pressure control and syncope |
| Hypertension |
| Atherosclerosis |
| Cardiac |
| Myocyte hypertrophy |
| Left ventricular hypertrophy |
| Arrhythmia |
| Psychogenic heart disease |
| Renal |
| Renal artery vasoconstriction |
| Sodium and fluid retention |
| Microalbuminuria |
| RAAS activation |
| Metabolic |
| Insulin resistance |
| Dyslipidemia |
RAAS: renin-angiotensin-aldosterone system.
There is growing evidence that the deleterious effects on blood vessels and myocardium of an overactive SNS are independent of increased BP.30 Chronic SNS activation without an increase in BP can cause hypertrophy and proliferation of vascular smooth muscle cells as well as having a direct trophic effect on cardiac myocytes, increasing left ventricular (LV) mass and wall thickness,30 while BP reduction after catheter-based RDN has been shown to lead to a significant reduction in LV mass and improvement in diastolic function.31
These structural changes in the myocardium and the direct effects of an overactive SNS contribute to the high incidence of arrhythmias commonly seen in patients with hypertension.32
The link between mental stress, psychiatric illness and cardiovascular disease, although best established for heart disease consequential to acute mental stress and depressive illness, has been much more difficult to establish.26 Acute mental stress can trigger sympathetic outflow to the heart and adrenal secretion of epinephrine. In patients with pre-existing atherosclerosis, not only can increased epinephrine cause ventricular arrhythmias (especially in the presence of coronary artery stenosis), but the attendant BP surge can fissure coronary plaques and promote platelet aggregation, predisposing to thrombosis.26 Takotsubo (stress) cardiomyopathy is a good example of extreme acute activation of cardiac sympathetic outflow to the heart,26 as are panic attacks accompanied by coronary spasm and cardiac arrhythmias.33 In patients with depressive illness, chronic cardiac sympathetic outflow is at almost the same level as seen in patients with heart failure, and is accepted as a primary cause for heart disease, associated with a worse prognosis.
The sympathetic nervous system and atherosclerosisThe pivotal role of endothelial impairment in the development of atherosclerosis and in future cardiovascular risk is well established. Less known is the interaction between the SNS and endothelial function. Virtually all cardiovascular risk factors and diseases in which increased adrenergic drive is involved are also characterized by endothelial dysfunction.34 Nitric oxide (NO), one of the main mediators of endothelial function, is also an important neurotransmitter, involved in the autonomic regulation of cardiovascular function, and acts as a sympathoinhibitory substance within the central nervous system.35 Acute and chronic increases in SNS activity, through endothelial dysfunction and endothelial cell damage, have been shown to contribute to the subsequent development of atherosclerosis.26,30,34
SNS overactivity has also been linked to the development of metabolic disturbances such as insulin resistance and dyslipidemia.36 Not only can increased SNS activity in itself lead to insulin resistance, particularly in hypertensive patients,36 but also elevated circulating insulin levels due to insulin resistance in obese patients can precipitate an increase in SNS activity, leading to hypertension.30,36,37 There is abundant evidence that statins, through their numerous pleiotropic effects, reduce and even normalize excessive SNS activity, improving LV function and arterial baroreflex sensitivity.26,30,34
Sympathetic nervous system function and heart failureThe observation that norepinephrine concentrations were reduced in the failing heart suggested the existence of sympathetic denervation,38 despite the increased concentrations in peripheral venous plasma commonly found in patients with heart failure,39 indicating overall increased SNS activity except in the heart. Later studies confirmed very high levels of norepinephrine spillover from the heart in heart failure, up to 50 times the normal range in untreated patients,26 demonstrating high sympathetic tone in the failing heart. This was later explained by a reduction in the concentration of beta-1 adrenoreceptors in the failing myocardium, due to downregulation of these receptors by increased sympathetic activity in the failing heart.26
This increased sympathetic activity in the peripheral circulation and kidneys leads to adverse effects, causing vasoconstriction, increasing cardiac work, and promoting sodium retention and ventricular overfilling. The strong link between the level of sympathetic activity in heart failure, progressive ventricular deterioration, the development of ventricular arrhythmias, sudden death and reduced survival26 provided the rationale for the subsequent use of beta-blockers in heart failure.40
Sympathetic nervous system function and essential hypertensionA well-known consequence of an overactive SNS is an increase in BP. Previous studies showed not only that sympathetic outflow to the kidneys was increased, but that the extent of the outflow was also related to the degree of essential hypertension.20,22,26,41 Regional measurements of norepinephrine spillover to the kidneys support this concept, and indicate that more than 50% of cases of essential hypertension present significant sympathetic hyperactivation.42 Renal sympathetic nerve activity is pivotal in the pathogenesis of essential hypertension, through its influence on renin release, sodium and water excretion, peripheral vasoconstriction, cardiac contraction and venous capacitance.26
The safety and efficacy of BP lowering achieved recently with RDN has led to renewed interest in the role of sympathetic activity in the pathogenesis of essential hypertension. Correct identification of sympathetic hyperactivation in patients with essential hypertension can lead to better selection of patients for RDN treatment.
Resistant hypertensionData from the PAP study involving 5023 adult patients showed that the prevalence of hypertension in Portugal was 42%, of whom only 46.1% were aware of the fact, 39% were taking medication and only 11.2% had BP values below the recommended thresholds.43
Not all patients with uncontrolled hypertension are considered to be resistant, as there are several factors that can contribute to lack of control, including inadequate treatment regimens (type and/or dosage of drugs), poor adherence to medical therapy, and undetected secondary causes of hypertension. A diagnosis of resistant hypertension should therefore only be made after ruling out other factors.
Resistant hypertension has been defined as BP values above 140/90 mmHg (or >130/80 mmHg in patients with diabetes or chronic kidney disease) in patients treated with three or more antihypertensive drugs at appropriate doses, including if possible a diuretic.44 In an alternative definition, patients with target BP values can be considered to have resistant hypertension if they need to take at least four different antihypertensive drugs; this is known as controlled resistant hypertension.45 The prevalence of resistant hypertension has been reported as 5–30%,46–48 the figure varying according to the hypertensive population being studied, with higher percentages in cohorts from centers specializing in the treatment of hypertension compared to community-based cohorts.
At the present time, and in the light of the available clinical studies, patients considered to be good candidates for RDN should have more severe treatment-resistant hypertension, defined as office systolic BP of at least 160 mmHg (150 mmHg in type 2 diabetes).49–51
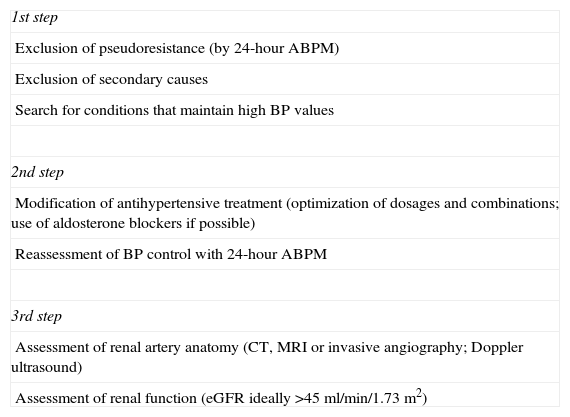
Renal denervationAssessment of a potential candidate for RDN should follow several steps (Table 2) designed to select candidates expected to benefit from this intervention. According to a recent European Society of Hypertension (ESH) position paper on RDN,52 it is recommended that patients should undergo careful assessment in centers that have considerable experience in dealing with hypertension (ideally ESH excellence centers).
Evaluation of patients with resistant hypertension considered to be potential candidates for renal denervation.
| 1st step |
| Exclusion of pseudoresistance (by 24-hour ABPM) |
| Exclusion of secondary causes |
| Search for conditions that maintain high BP values |
| 2nd step |
| Modification of antihypertensive treatment (optimization of dosages and combinations; use of aldosterone blockers if possible) |
| Reassessment of BP control with 24-hour ABPM |
| 3rd step |
| Assessment of renal artery anatomy (CT, MRI or invasive angiography; Doppler ultrasound) |
| Assessment of renal function (eGFR ideally >45 ml/min/1.73 m2) |
ABPM: ambulatory blood pressure monitoring; BP: blood pressure; CT: computed tomography; eGFR: estimated glomerular filtration rate; MRI: magnetic resonance imaging.
The first step should be to exclude pseudoresistance, secondary causes of hypertension and conditions that maintain high BP values. Pseudoresistance can be excluded by 24-hour ambulatory BP monitoring, which is recommended not only for pre-RDN assessment but as good practice in the assessment of all patients with hypertension.5,53 In patients with severe resistant hypertension considered for RDN, the initial assessment should also include investigation of possible secondary causes of hypertension, including primary aldosteronism, renal artery stenosis, pheochromocytoma, Cushing's disease, hyperparathyroidism and aortic coarctation, although some of these are very uncommon.44 Some conditions that can maintain high BP values should also be treated when possible, such as severe obesity, high salt and alcohol intake, concomitant use of drugs that raise BP and the presence of obstructive sleep apnea.
The second step should be the optimization of antihypertensive treatment, including the use of diuretics and aldosterone blockers, optimization of dosages and combinations, and reassessment of BP control with 24-hour ambulatory BP monitoring (ABPM).
The third step should be assessment of renal artery anatomy, as there are relative contraindications for RDN related to the number of renal arteries (multiple main arteries) and their diameter (ideally >4 mm) and length (ideally >20 mm) as well as eGFR, which should be above 45 ml/min/1.73 m2. Some of these are considered contraindications to RDN, because they were excluded from RDN trials, but are regarded in clinical practice as relative contraindications; some of these patients have been treated with RDN and included in small studies and registries.54–56
Experimental studies on renal denervationThe finding that renal sympathetic activity is increased in spontaneously hypertensive rats, the animal model most often used in investigation of essential hypertension, has shone light on the role of the renal SNS in the pathogenesis of hypertension.57 In an experimental model of hypertension and obesity in dogs subjected to a high-fat diet, RDN not only prevented the appearance of hypertension but also increased urinary sodium excretion by 50%.58 In another animal model of chronic renal failure, sympathectomy prevented hypertension and was associated with decreased adrenergic activity in the hypothalamic nuclei.59
Renal lesions induced by injection of phenol cause a sustained rise in BP and norepinephrine release by the hypothalamus without changing eGFR. RDN of these animals prevented the rise in BP.60 In different animal models, the effects of RDN have consistently shown the important role of the renal SNS in the pathophysiology of hypertension.
In humans, surgical sympathectomy lowers high BP and improves the cardiovascular prognosis of patients with severe hypertension.7–9,61–63 Its poorly tolerated side effects, which include severe orthostatic hypotension, anhidrosis, intestinal disturbances and sexual dysfunction, and its high surgical risk, have led to the technique being abandoned. Nevertheless, it has proved the importance of the SNS in BP control beyond doubt.
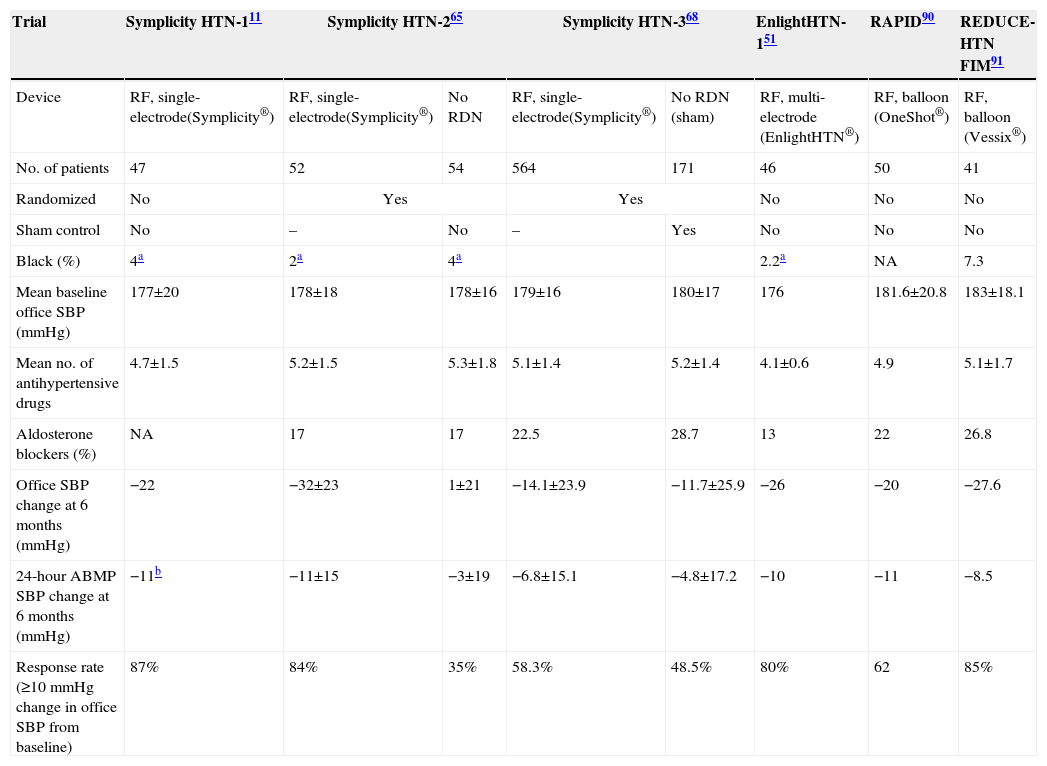
Clinical studies on percutaneous sympathetic renal denervationSymplicity HTN-1,11 a proof-of-principle study, was the first to evaluate RDN in patients with severe resistant hypertension. One year after the procedure, mean office BP fall was 27 mmHg systolic and 17 mmHg diastolic, and was maintained until 24 months of follow-up, with 13% non-responders. A subgroup analysis assessing renal and systemic sympathetic activity showed a 47% reduction in renal norepinephrine spillover in these patients.64
In the Symplicity HTN-2 multicenter clinical trial,64,65 106 patients with severe resistant hypertension under medication were randomized to optimal antihypertensive medical therapy alone or to RDN plus optimal antihypertensive medical therapy. The primary endpoint was change in office BP at six-month follow-up. A significant fall in BP was observed in patients who underwent RDN: −32 mmHg in systolic BP and −12 mmHg in diastolic BP (p<0.01) compared to an increase of 1 mmHg in systolic BP and no change in diastolic BP (p=NS) in patients under optimal medical therapy alone at six months following RDN. A subgroup analysis of 24-hour ABPM data revealed a similar pattern, a fall of 11 mmHg in systolic BP and 7 mmHg in diastolic BP in the RDN group (p<0.001) compared to a fall of 3 mmHg and 1 mmHg on medical therapy alone (p=NS). The magnitude of the difference in BP fall between RDN and optimal medical therapy alone was maintained at 12-month follow-up.64
Alongside its efficacy, RDN was a safe procedure. In both studies, only minor vascular complications occurred, mainly at the puncture site: hematomas and pseudoaneurysms (four patients), one renal artery dissection during the diagnostic procedure, successfully treated with a stent, and no major complications. Regarding renal function, there were no significant changes in eGFR during follow-up.49,66
The recently published EnlightHTN I trial51 also revealed a significant fall in both office BP and mean 24-hour ABPM values at six months, with a good safety profile.
The published data indicate that RDN has an excellent short-term safety profile, although data on the long-term risk of renal artery stenosis are lacking. The results of the Symplicity trials and EnlightHTN I are certainly promising, but their open design meant that bias in BP measurements, or even the extent of the placebo effect in treated patients, could not be properly addressed. Some of these limitations were addressed in the recently published randomized SYMPLICITY HTN-3 trial.67,68 This was the first blinded sham-controlled study of RDN for treatment of resistant hypertension. The primary efficacy endpoint was the mean change in office systolic BP from baseline to six months in the RDN arm (n=364) compared to the control arm (n=171). At six-month follow-up, there was a difference of 2.39 mmHg in the change in systolic BP (−14.13±23.93 mmHg in the RDN arm vs. −11.74±25.94 mmHg in the sham procedure arm), which did not reach statistical significance (95% confidence interval [CI]: −6.89 to 2.12, p=0.26).
The secondary efficacy endpoint was the change in mean 24-hour ambulatory BP at six months. A statistically non-significant difference of 1.96 mmHg (95% CI: −4.97 to 1.06, p=0.98) was seen at six months (−6.75±15.11 mm Hg in the RDN arm vs. −4.79±17.25 mm Hg in the control arm).
The rate of major adverse events at six months was 4% in the RDN arm vs. 5.8% in the control arm (p=0.37). The primary safety endpoint (a composite of major adverse events) rate was 1.4% in the RDN arm, less than the pre-specified objective of 9.8%, reaching statistical significance (p<0.001).
These conflicting results between Symplicity HTN-368 and the previous Symplicity trials, HTN-111 and HTN-265 (Table 3), may be related to a different and more rigorous design adopted in HTN-3, a different study population, more aggressive antihypertensive medication, and the requirement that no changes in antihypertensive medication could be made in the six months after the procedure. There was also potential for procedural variability due to the large number of centers involved in the HTN-3 study and a low case load per operator, each performing only three procedures on average (most performed their first and only RDN procedure for the trial). The inclusion for the first time of a large proportion of African-American patients (24.8% of the RDN arm and 29.2% of the control arm), a population known to be resistant to RAAS blockers, could have had a negative impact on the efficacy of RDN; a subgroup analysis revealed a statistically significant difference favoring the RDN arm in non-African-American patients. Overall antihypertensive medication was more intensive than in previous studies, probably reflecting the more severe hypertensive patients included. Regression to the mean may also at least partially explain the differences.69 The presence for the first time of a sham procedure in the control arm (renal angiography was performed in all patients before randomization) may have diluted the expected placebo effect favoring the treated group.
Studies of renal denervation.
| Trial | Symplicity HTN-111 | Symplicity HTN-265 | Symplicity HTN-368 | EnlightHTN-151 | RAPID90 | REDUCE-HTN FIM91 | ||
|---|---|---|---|---|---|---|---|---|
| Device | RF, single-electrode(Symplicity®) | RF, single-electrode(Symplicity®) | No RDN | RF, single-electrode(Symplicity®) | No RDN (sham) | RF, multi-electrode (EnlightHTN®) | RF, balloon (OneShot®) | RF, balloon (Vessix®) |
| No. of patients | 47 | 52 | 54 | 564 | 171 | 46 | 50 | 41 |
| Randomized | No | Yes | Yes | No | No | No | ||
| Sham control | No | – | No | – | Yes | No | No | No |
| Black (%) | 4a | 2a | 4a | 2.2a | NA | 7.3 | ||
| Mean baseline office SBP (mmHg) | 177±20 | 178±18 | 178±16 | 179±16 | 180±17 | 176 | 181.6±20.8 | 183±18.1 |
| Mean no. of antihypertensive drugs | 4.7±1.5 | 5.2±1.5 | 5.3±1.8 | 5.1±1.4 | 5.2±1.4 | 4.1±0.6 | 4.9 | 5.1±1.7 |
| Aldosterone blockers (%) | NA | 17 | 17 | 22.5 | 28.7 | 13 | 22 | 26.8 |
| Office SBP change at 6 months (mmHg) | −22 | −32±23 | 1±21 | −14.1±23.9 | −11.7±25.9 | −26 | −20 | −27.6 |
| 24-hour ABMP SBP change at 6 months (mmHg) | −11b | −11±15 | −3±19 | −6.8±15.1 | −4.8±17.2 | −10 | −11 | −8.5 |
| Response rate (≥10 mmHg change in office SBP from baseline) | 87% | 84% | 35% | 58.3% | 48.5% | 80% | 62 | 85% |
ABPM: ambulatory blood pressure monitoring; NA: not available; RDN: renal denervation; RF: radiofrequency; SBP: systolic blood pressure.
Puzzling findings in Symplicity HTN-3 include the smaller decrease in office systolic BP from baseline to six months in the RDN arm, about half that observed in the Symplicity HTN-2 RDN group, despite similar baseline BP in the two studies, raising doubts as to whether RF energy was properly delivered. A larger decrease in BP was also observed in the HTN-3 control group compared to the much smaller decrease in the HTN-2 control group. These findings raise the question of whether a less effective denervation procedure allied to more aggressive medical therapy could have played a major role in the HTN-3 results.
The fact that there was no measurement to confirm that the renal nerves were in fact denervated by the procedure, because there is no test that can be easily performed in a large trial, is a major limitation to this and to almost all of these trials.
While the Symplicity HTN-3 follow-up will continue as planned for up to five years, the fact that many patients crossed over from the control arm to the RDN arm at six months will make it more difficult to draw significant conclusions concerning the long-term clinical results of RDN therapy and to assess the placebo effect over time.
Nevertheless, the Symplicity HTN-3 trial is a landmark in the development of RDN treatment, signaling the start of its reflection phase, in which new hypotheses generated by this trial can be addressed.
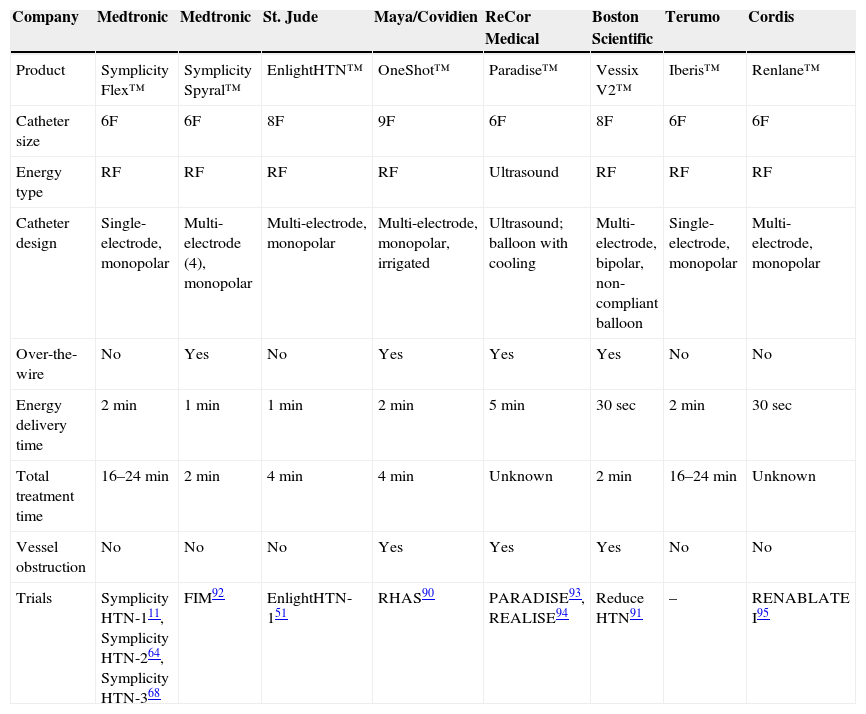
Renal denervation: new devicesThe high expectations and enthusiasm created in the medical device industry has led many companies to develop new or improved technical solutions for RDN, some of which are commercially available (Table 4).
Sympathetic renal denervation devices.
| Company | Medtronic | Medtronic | St. Jude | Maya/Covidien | ReCor Medical | Boston Scientific | Terumo | Cordis |
|---|---|---|---|---|---|---|---|---|
| Product | Symplicity Flex™ | Symplicity Spyral™ | EnlightHTN™ | OneShot™ | Paradise™ | Vessix V2™ | Iberis™ | Renlane™ |
| Catheter size | 6F | 6F | 8F | 9F | 6F | 8F | 6F | 6F |
| Energy type | RF | RF | RF | RF | Ultrasound | RF | RF | RF |
| Catheter design | Single-electrode, monopolar | Multi-electrode (4), monopolar | Multi-electrode, monopolar | Multi-electrode, monopolar, irrigated | Ultrasound; balloon with cooling | Multi-electrode, bipolar, non-compliant balloon | Single-electrode, monopolar | Multi-electrode, monopolar |
| Over-the-wire | No | Yes | No | Yes | Yes | Yes | No | No |
| Energy delivery time | 2 min | 1 min | 1 min | 2 min | 5 min | 30 sec | 2 min | 30 sec |
| Total treatment time | 16–24 min | 2 min | 4 min | 4 min | Unknown | 2 min | 16–24 min | Unknown |
| Vessel obstruction | No | No | No | Yes | Yes | Yes | No | No |
| Trials | Symplicity HTN-111, Symplicity HTN-264, Symplicity HTN-368 | FIM92 | EnlightHTN-151 | RHAS90 | PARADISE93, REALISE94 | Reduce HTN91 | – | RENABLATE I95 |
RF: radiofrequency.
From predictable improvements of the original procedure to out-of-the-box ideas, many innovations are being integrated in the new designs. These include alternative mechanisms of action, like ultrasound catheters and balloons with microinjection systems to deliver neurotoxins. Simultaneously activated multi-electrodes not only significantly shorten procedural time but also increase reproducibility, ensuring that all quadrants are adequately denervated, while radial access reduces access site vascular complications, and manipulating renal catheters by a craniocaudal approach is generally less challenging and safer. Pain control is also a challenge, as electric current, tissue burning, and nerve damage, although essential components of the procedure, all cause discomfort. The more recent radiofrequency catheters with bipolar electrodes reportedly reduce discomfort due to a significantly smaller electric field during activation, but this is not yet clinically proven; the absence of an acute procedural efficacy endpoint is still a major limitation. Procedural success is difficult to determine and to correlate with BP response. Efforts are being made to find a biomarker or physiological test that indicates acute RDN success.
Sympathetic renal denervation: potential future indicationsThe overall decrease in SNS drive through RDN may be a valid alternative in clinical scenarios characterized by sympathetic hyperactivity other than resistant hypertension. A few of these alternative applications have already been explored and show promising results.
The association between heart failure and increased sympathetic drive is well known. Interestingly, cardiac and renal norepinephrine spillover is more closely associated with mortality than circulating catecholamine concentrations, although both are associated with worse outcomes.16,70 This suggests that reducing norepinephrine spillover from the kidney could have beneficial symptomatic and prognostic effects.71,72
In animal models, RDN after myocardial infarction led to improvement in sodium excretion,73 increased cardiac output, improved renal blood flow,74 and down-regulation of angiotensin AT1 receptors mediating maladaptive responses.75 In a multicenter study of patients with resistant hypertension treated by RDN with anatomical and functional myocardial parameters assessed by MRI, a subgroup of patients with LV dysfunction had significantly increased ejection fraction and circumferential strain.76
The REACH pilot study in heart failure provided evidence that RDN improved six-minute walk distances without affecting BP (mean 120 mmHg at baseline).77 Other ongoing clinical trials will provide further evidence on the potential of RDN to influence the course and outcome of heart failure. Type 2 diabetes and insulin resistance are other conditions that have a strong association with resistant hypertension. Half of resistant hypertension patients are considered to be insulin resistant, increasing the risk for type 2 diabetes, and since insulin resistance is dependent on sympathetic activity it appears likely that it could also be a target for RDN.78,79 In a pilot study, along with reducing BP, RDN improved fasting glucose, insulin, and C-peptide concentrations, as well as homeostasis model assessment-insulin resistance indices in patients with resistant hypertension and metabolic disease, suggesting that RDN might improve diabetic status in these patients.80 Witkowski et al. showed a decline in glycated hemoglobin concentrations after RDN.81
The association between obstructive sleep apnea and resistant hypertension is well known.81 In 2011 Witkowski et al. published a pilot study on the effect of RDN in 10 patients with resistant hypertension and obstructive sleep apnea. At six months there was an improvement in apnea-hypopnea indices.81 In an experimental model, it has been shown that RDN reduces post-apneic BP rise, renal hypoperfusion during apnea and RAAS activation in the kidney.82,83 The value of these findings is still controversial and confirmatory studies are needed.
In an animal model84 of obstructive sleep apnea and induced atrial fibrillation (AF), RDN decreased the atrial refractory period and AF recurrence,82 providing better rate control.85 In a pilot trial, patients with resistant hypertension and symptomatic paroxysmal or persistent atrial fibrillation refractory to ≥2 antiarrhythmic drugs were randomized to pulmonary vein isolation alone or associated with RDN. At 12-month follow-up 69% of patients treated with RDN were AF-free, compared to 29% of those treated with pulmonary vein isolation only.86 These experimental findings indicate the potential usefulness of RDN in AF treatment.
The evidence for the fundamental role of sympathetic activity in ventricular arrhythmias is overwhelming. In an animal model of ischemia/reperfusion arrhythmias, RDN decreased the occurrence of ventricular arrhythmias/fibrillation and attenuated the rise in LV end-diastolic pressure during LV ischemia without influencing infarct size, changes in ventricular contractility, BP or reperfusion arrhythmias.87 In a small case series involving patients with chronic heart failure and ventricular electrical storm, RDN reduced discharges from implantable cardioverter-defibrillators and ventricular ectopies.88 Hoffmann et al.89 reported that RDN can be safely and effectively performed as an adjunct to cardiac catheter ablation, in a hemodynamically unstable patient with ventricular storm after ST-elevation myocardial infarction. Although these are early and preliminary findings, the underlying biological plausibility will certainly heighten interest in these potential future applications of RDN.
ConclusionsIt is now accepted that an overactive sympathetic system has a pivotal role in the pathophysiology of several diseases besides essential hypertension. Related conditions like depression, mental stress, hypertension, diabetes, obesity, sleep apnea, metabolic syndrome, ischemic heart disease, heart failure and chronic renal failure, all have a common link, the often neglected hyperactive SNS. In a new era with new tools to control and treat sympathetic hyperactivity, perhaps this system will finally receive the attention it deserves.
The inability to treat hypertension effectively is due in part to a lack of understanding of the fundamental mechanisms involved in BP control. There is a complex mixture of hormonal, neural and intrinsic factors, all acting together, over different time scales and with different feedback control pathways, and it seems unlikely that any of the current treatment approaches is actually targeting the factors that originally led to the rise in BP. Catheter-based RDN is a truly innovative approach to treat hypertension by changing sympathetic activity. In patients with resistant hypertension, the technique has significantly reduced BP as well as sympathetic nerve activity and norepinephrine spillover, with high safety levels. These achievements are well documented in several international multicenter trials and registries. Along with its proven efficacy in BP reduction, it has the potential to positively affect insulin resistance and diabetes, LV mass, proteinuria and arrhythmias, as indicated by various small proof-of-concept studies.
Nevertheless, there are still important issues that need to be addressed in the near future, like the impossibility of determining whether denervation was effective, what level of denervation is needed to achieve clinical success, which patients have an appropriate phenotype for RDN, and what endpoints should be used to define RDN success (merely BP reduction or reduction in target organ damage). Much needs to be done and will be in the coming years, but a new window has certainly been opened not only to address hypertension, but most importantly to address SNS dysfunction.
Conflicts of interestThe authors have no conflicts of interest to declare.